Anti-antibiotics
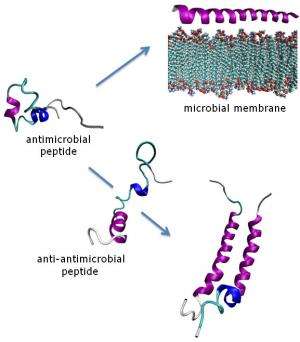
Antimicrobial peptides are natural antibiotics found in all multicellular organisms. These molecules are viewed as potential drug candidates in the post-antibiotic era because widespread microbial resistance against them has yet to emerge.
The use of these peptides in medicine may trigger the development of antimicrobial resistance in a fashion similar to that currently occurring with more conventional antibiotics such as methicillin and vancomycin. However, the mechanism of action of antimicrobial peptides, which target and rapidly destroy bacterial membranes, lends credibility to the view that this is unlikely to happen. To acquire resistance against such formidable agents, microorganisms would need to re-build their genetic apparatus, which is an extremely high price to pay.
In a recent study, a research team comprising scientists from NPL, University of Bristol, University of Edinburgh, University of Oxford and IBM explored the effect of peptide sequences designed to be antagonistic towards antimicrobial peptides. The research showed that these 'anti-antimicrobial' sequences interact with the antimicrobial peptides to form inert molecular complexes, known as helical oligomers.
The team then studied the structural and biological properties of these inert assemblies in biologically relevant environments, using a combination of spectroscopy, microscopy, bioassays and molecular dynamics simulations. The findings, reported in the Journal of Biological Chemistry, suggest that antagonistic sequences secreted by bacterial cells or expressed on their surfaces may cause efficient anti-antimicrobial responses. Although this remains to be shown in nature, this research offers a molecular rationale for these responses with potential implications for antimicrobial resistance.
Lloyd Ryan, an NPL research scientist who worked on the project, said:
"It is widely believed that antimicrobial peptides are powerful antibiotics that can overcome the microbial resistance we see nowadays in clinics. However, for us it was important to understand if a relatively simple mechanism of resistance is plausible against antimicrobial peptides before they are considered for use on an industrial scale."
More information: www.jbc.org/content/288/28/20162.long

















