Cells make costume changes for cardiac regeneration
(Medical Xpress)—If the heart following a heart attack is not sufficiently supplied with blood, heart tissue dies. In adult humans, the ability to heal itself is hardly developed. Scientists from the Max Planck Institute for Heart and Lung Research in Bad Nauheim, together with U.S. colleagues, have now observed in the embryo of the zebrafish that muscle cells migrate from the undamaged atrium into the ventricle and thus significantly contribute to regeneration. This could serve as the basis for novel therapeutic approaches.
If clinicians fail to reopen occluded coronary arteries after a heart attack within an appropriate time frame, the heart muscle is permanently damaged because of the long-term interrupted oxygen supply. The result is, among other things, a lifelong restriction of cardiac function, or even heart failure.
For many years, scientists worldwide have been searching for ways to stimulate the regeneration of damaged heart tissue. The working group of Didier Stainier from the Max Planck Institute for Heart and Lung Research has now, together with scientists from the University of San Diego, identified in zebrafish a novel mechanism, at which muscle cells from the atrium actively migrate into damaged parts of the heart muscle in the ventricle, thus forming new ventricular tissue.
For their study, the Max Planck researchers used genetically modified fish larvae, in which the targeted muscle cells of the heart chamber were destroyed by the administration of a substance. This was done at a point in time, at which the heart was already functional and active. The damage of the muscle was followed by a reduction in heart function and ventricular size.
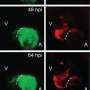
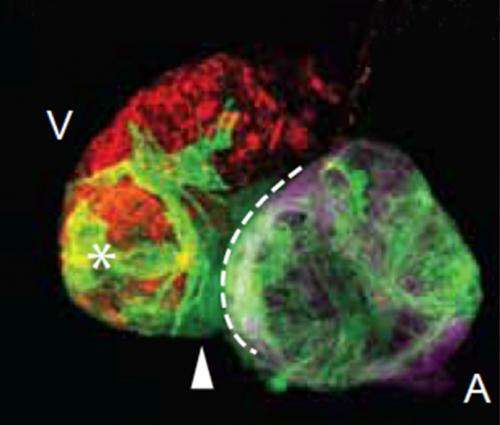
To monitor the behaviour of the different cell types, the heart muscle cells were furthermore altered by genetic engineering in such a way that cells from the atrium and the ventricle lit up differently. "In this way we were able to track, the behaviour of the individual cell types continuously in a confocal microscope," explains Didier Stainier, the director of the department "Developmental Genetics" at the MPI.
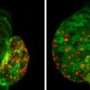
"A few hours after ablation only few red cells were remaining in the ventricle. Furthermore, it had been significantly shrunk, both references to the death of muscle cells," says Stainier. Just 24 hours later, much more fluorescing cells were back, indicating the initiation of cell division activity of surviving cells within the ventricle.
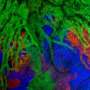
Such a regeneration ability in zebrafish has been known for a long time. The real surprise was the fact that increasingly green glowing cells from the atrial muscle cells migrated into the ventricle. A few days after the injury of muscle tissue, large parts of the ventricle were glowing green.
In further studies, the scientists then found clear evidence of a so-called transdifferentiation of cells: The muscle cells of the atria of the heart of the fish lost their characteristic properties and subsequently transformed into ventricular cells in the course of regeneration. With the progression of heart regeneration, these cells were permanently installed in the muscle tissue and made their contribution to the restoration of cardiac function.
The Max Planck researchers in their study see potential for future therapy. "Although, a atrial cell population is known to be present in humans with comparable properties, it is questionable whether the human heart possesses a similar self-healing ability," says Stainier. One solution might be, however, using gene therapy to stimulate such reprogramming of cells and thus to strengthen the self-healing abilities of the heart.
More information: Zhang, R. et al. In vivo cardiac reprogramming contributes to zebrafish heart regeneration, Nature. 2013 Jun 27; 498(7455):497-501. doi: 10.1038/nature12322