Just a few cell clones can make heart muscle
Just a handful of cells in the embryo are all that's needed to form the outer layer of pumping heart muscle in an adult zebrafish.
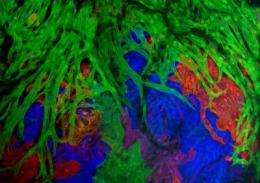
Researchers at Duke University Medical Center used zebrafish embryos and careful employment of a new technique that allows for up to 90 color labels on different cells to track individual cells and cell lines as the heart formed.
The scientists were surprised by how few cells went into making a critical organ structure and they suspect that other organs may form in a similar fashion, said Kenneth Poss, Ph.D., professor in the Duke Department of Cell Biology and Howard Hughes Medical Institute.
The study appears online in Nature on April 25.
"The most surprising aspect of this work is that a very small number of cardiomyocytes (heart muscle cells) in the growing animal can give rise to the thousands of cardiomyocytes that form the wall of the cardiac ventricle," said Vikas Gupta, lead author, who is in the Duke Medical Scientist Training Program for M.D. and Ph.D. degrees.
Gupta found that about eight single cells contributed to forming the major type of heart muscle in the wall of the zebrafish heart -- and just one or two cells could create anywhere from 30 to 70 percent of the entire ventricular surface.
"Clonal dominance like this is a property of some types of stem cells, and it's a new concept in how to form an organ during development," Poss said.
Another surprise was the way the patches of cloned cells formed muscle.
"It was completely unexpected," Gupta said. "I thought the wall would simply thicken in place, but instead there was a network of cells that enveloped the ventricle in a wave. It was as if a cell at your shoulder grew a thin layer of new cells down your arm surface."
Gupta said this opens an area for investigation to see whether or not a process like this repeats in the hearts of mammals, and perhaps in other internal organs.
Poss said the cell clones appear to have the ability to cover as much of the ventricular surface as possible before other cells start appearing and growing at the surface.
"Our suspicion is that the muscle cells that initiate large clones are not much different from other muscle cells – they just get to the surface of the heart first," Poss said.
They used the analogy of a sperm getting to the egg first, among all the millions of possible sperm cells.
Poss said the manner in which these muscle cells envelope the heart could lead to new therapies.
"Researchers may be able to channel this developmental process to help damaged hearts or failing hearts to grow muscle that will reinforce the ventricular walls," he said. "Someone who's had a heart attack would want this ability to generate new muscle to cover a scar naturally, and it's attractive to think that the help might come from a small number of muscle cells within a population."
The color-label technique was originally developed by other biologists and was critical to allow the researchers to track heart cell populations.
"You can label individual cells very early in an embryo with a permanent color and those cells and their progeny will keep that color," Poss said. "You can learn what an individual cardiomyocyte did, and its neighbor, and that cell's neighbor and so on, until you've covered much of the whole ventricle of the developing zebrafish."
Poss said it makes sense that this growth process works by a gradual layering process, especially for the heart. "It's speculative, but for the heart to maintain circulation in a relatively slowly growing animal, a process like this to build the heart might be a way of gradually increasing its circulatory strength to keep up."