LITE illuminates new way to study the brain
For core faculty member Feng Zhang's lab, nature offers up the raw ingredients needed to control brain cells and understand the brain's wiring. Since joining the Broad Institute and McGovern Institute for Brain Research in 2011, Zhang has recruited neuroscientists, engineers, computer scientists, and others from diverse backgrounds who are interested in developing and optimizing the tools needed to understand the brain. The researchers have turned to bacteria for proteins that can be engineered to bind to precise locations in the genome, and to plants and algae for proteins that are activated by light. Now, Zhang's research team has brought discoveries from both areas of study together to create an expansive toolset for precisely controlling and testing the function of genes and other genetic influences in the brain.
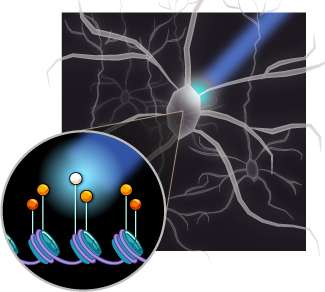
The new research, published online in Nature, builds upon the Zhang lab's work with transcription activator-like effectors (TALEs), highly customizable factors that can be engineered to bind anywhere in the genome. TALEs offer an anchor point from which to tweak and modify the genome. In the latest experiments, the team has attached a protein that's only active when light is present, giving them further control over not only where but when to turn on or off molecular machinery.
"These tools combine the DNA targeting abilities of TALEs with light-sensitive proteins," said co-first author Silvana Konermann, a graduate student in Zhang's group and a neuroscientist by training. "That means that we can affect a process at a specific locus in the genome but with light: we can either activate or repress transcription, and we can also modify the epigenetic state."
The new tools, known as LITEs (light-inducible transcriptional effectors), give researchers unprecedented, fine-tuned control to test genes of interest. By activating transcription, they can turn on the readout of a gene, much like flipping on a printer to churn out copies of a manuscript. Conversely, they can repress transcription, turning off, or at least slowing down, the output. LITEs can help researchers get a handle on not only genes, but also the machinery that surrounds them, responsible for controlling where and when genes are turned on. Having the ability to test these epigenetic factors holds much promise for many fields of research, including the study of neuropsychiatric disease, cognition, cancer, and more.
"There's a growing amount of epigenomic data," said co-first author Mark Brigham, a bioengineering graduate student in Zhang's group. "Those data are generating lots of hypotheses about the role of epigenetics in disease and in an organism. Right now, we have very few tools to take those data and hypotheses and test them in a controlled manner. In this work, we've expanded the toolbox significantly. There's a great deal of work that still needs to be done, but we've kicked off this idea and given people a place to start."
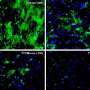
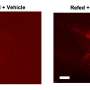
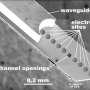
The Nature paper describes the team's efforts to optimize and validate LITEs, both in cells in the dish and in the brains of mice. To do so, the researchers injected viral particles containing LITEs into neurons. In the case of the mice, they then threaded a fiber optic cable into the brain, allowing them to deliver light directly into the cells. By turning the light on or off, the researchers can stop and start the activation or repression of a gene.
The technique of using light to control the activity of neurons, known as optogenetics, has been used in the past to control electrical signaling in specific neurons in the mouse brain. Researchers can use light to control the opening of ion channels, which allow electrical signals to flow within the brain. The current work looks at a different level of neural activity, giving researchers the ability to control genes instead of ion channels.
"When an animal is learning or exposed to a new environment, certain genes get transcribed," said Konermann. This process can unfold over hours. "When we file long-term memories, it's thought there is an epigenetic change. It's important for us to be able to target all three levels – electrical signaling, genes and transcription, and epigenetics – in order to better understand the brain's function."
In this work, the researchers measured the effects of LITEs by measuring gene activity, but in the future, they intend to measure subtle behavioral changes in mice, resulting from the contributions of a single gene.
"You can imagine a lot of potential uses for this technique in many areas," said Brigham. "There are applications in cancer biology because we know that the roots of tumor development not only involve genetic changes, but the dysregulation of epigenetic states. Making changes in a spatially and temporally confined manner allows for greater precision."
The team plans to collaborate closely with researchers at MIT, the Broad's Stanley Center, and elsewhere who are studying the brain. They also anticipate that researchers studying other aspects of genomics and biomedicine will find the toolbox useful.
More information: Nature DOI: 10.1038/nature12466