New treatment for brittle bone disease found

A new treatment for children with brittle bone disease has been developed by the University of Sheffield and Sheffield Children's Hospital.
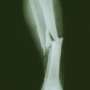
The study of the new treatment for children with the fragile bone disease Osteogenesis Imperfecta was published this week in the world's leading general medical journal, The Lancet.
This is the first study to clearly demonstrate that the use of the medicine risedronate can not only reduce the risk of fracture in children with brittle bones but also have rapid action - the curves for fracture risk begin to diverge after only 6 weeks of treatment.
Nick Bishop, Professor of Paediatric Bone Disease at the University of Sheffield, said: "We wanted to show that the use of risedronate could significantly impact on children's lives by reducing fracture rates - and it did.
"The fact that this medicine can be given by mouth at home (other similar medicines are given by a drip in hospital) makes it family-friendly."
The study, funded by the Alliance for Better Bone Health, trialled children with Osteogenesis Imperfecta aged 4-15 years and showed that oral risedronate reduced the risk of first and recurrent clinical fractures and that the drug was generally well tolerated.
More information: To read the published article in The Lancet, titled Risedronate in children with osteogenesis imperfecta: a randomised, double-blind, placebo-controlled trial, visit The Lancet. www.thelancet.com/journals/lan … rticle/PIIS0140-6736%2813%2961091-0/fulltext