Carbon monoxide could hold promise of effective preeclampsia treatment, prevention
Preeclampsia (PE) is a high blood pressure disorder that occurs during pregnancy and which can cause illness or death for the fetus and mother-to-be. There is currently no cure except to deliver the fetus, perhaps prematurely, or remove the placenta, a key organ that binds the pair. Women who smoke during pregnancy have been found to have as much as a 33 percent lower rate of preeclampsia for reasons that are unclear. A new study using an animal model to mimic key effects of PE in humans, and led by Graeme Smith of Queen's University, Canada, may help explain the lower incidence of PE in some smokers. The study also suggests a therapy that could offer similar protection against PE with none of smoking's well-known risks.
The article is entitled "Chronic Carbon Monoxide Inhalation During Pregnancy Augments Uterine Artery Blood Flow and Uteroplacental Vascular Growth in Mice." It appears in the Articles in PresS edition of the American Journal of Physiology – Regulatory, Integrative, and Comparative Physiology, published by the American Physiological Society. It is available online.
Background
Research has found that, compared with that of healthy pregnant women, the breath of women who are diagnosed with PE contains significantly lower levels of carbon monoxide (CO), one of the combustible elements in cigarettes and which is not present in smokeless tobacco. Although toxic at high levels, low levels of CO are produced naturally by the body and have positive effects.
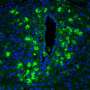
This study, the first to look at the effects of CO on the placenta in mice, found that exposing the animals to inhalable CO increased blood flow and vascular growth in the developing placenta, created more, bigger, and stronger connections to the mother's uterus, a process that provides more oxygen and nutrients to the fetus. And, since PE is a multifaceted disorder that begins with abnormal placental development before progressing to maternal disease, the results also suggest the possibility of preventing or treating early PE in humans with controlled CO dosages.
Methodology
Beginning on gestation day, pregnant female mice were placed in a sealed chamber with as much food and water as they wanted and exposed to CO, either chronically or acutely twice during gestation, at levels that allowed mouse CO blood levels to reach levels comparable to a one-pack-per-day female smoker. Maternal weight, live fetuses, fetal resorptions, and implantation sites were compared to control mice. Doppler analysis was performed on day 5 (baseline), 10 (when placental structure has formed and blood begins to cross the placenta to the fetus), and 14 (when blood velocity is well-established and fetal vasculature begins). Mice were then anesthetized and a cast was made of the entire uterus and blood vessels, then mounted and three-dimensionally imaged by micro-CT.
Results
Exposure to CO either continuously or in specific doses did not affect maternal weight (used as a measure of health) or litter size. Compared to controls, CO exposure did lead to increased vessel diameter, a significant increase in the number of radial artery branches, and significantly higher maternal blood flow.
Importance of the Findings
The study, according to Dr. Smith, confirms for the first time in vivo that CO has a beneficial effect at the placental level. It also provides an explanation for the lower incidence of PE among smokers, whose CO levels are higher. Positive changes in placental development in mice breathing additional CO – and earlier studies in the Smith lab showing these levels of CO had no negative effect on the developing fetus—suggest a potential role for CO inhalations in preventing PE in patients at risk (perhaps because of severe PE in a previous pregnancy) or in attenuating early stage PE, preventing abnormal placental development from progressing to maternal disease.
Study Team
In addition to Dr. Smith, the study team consisted of Carolina Venditti, Casselman and Malia SQ Murphy, also of Queen's University; S. Lee Adamson of Mount Sinai Hospital, Toronto, and the University of Toronto; and John G. Sled of the Hospital for Sick Children's Mouse Imaging Centre and the University of Toronto.