September 24, 2013 report
Decoy FGFR3 protein appears to prevent dwarfism in mice
(Medical Xpress)—A team made up of researchers from several institutions in France has found that a decoy protein injected into mice, appears to prevent the development of dwarfism. In their paper published in the journal Science, the team describes how they used a recombinant protein therapeutic approach with a soluble form of a human protein to counteract the gene mutation responsible for dwarfism in mice.
Dwarfism is a condition where people have short arms and legs relative to their torso and head. In addition to causing short stature, it has been found to cause spinal and breathing problems. Currently, there is no cure for the condition. In this new effort, the researchers in France are reporting that they've made major headway in preventing it from happening—at least in mice.
Prior research has revealed that most types of dwarfism are caused by a mutation in the FGFR3 gene—it leads to receptors on the surface of chondrocytes (cartilage cells) becoming hyperactive, preventing the proper formation of bone. To prevent the chondrocytes from becoming hyperactive, the researchers developed a soluble form of a protein to act as a decoy—they called it sFGFR3—it bonds with growth factors in bone cells instead of with those generated by the mutant FGFR3 gene protein.
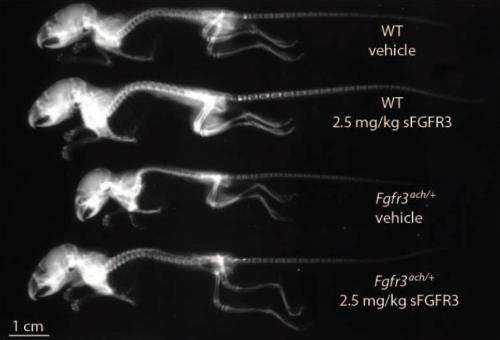
The result, the team reports is the prevention of dwarfism in test mice (that had a condition similar to achondroplasia—the most common form of dwarfism in humans) that were injected with the protein twice a week for three weeks. They report also that other problems associated with dwarfism disappeared as well and that the treated mice gave birth to normal mice after reaching adulthood, showing that the skeletal structures of the mice were sufficient for delivery.
So optimistic are the researchers about their results, they are predicting that their protein will be ready for clinical trials very soon and that the protein could conceivably be ready for delivery to patients as soon as three to five years from now—effectively eradicating dwarfism in the future. They point out that the therapy will only work when given to children (pre-puberty) most likely, shortly after birth.
More information: S. Garcia, B. Dirat, T. Tognacci, N. Rochet, X. Mouska, S. Bonnafous, S. Patouraux, A. Tran, P. Gual, Y. L. Marchand-Brustel, I. Gennero, E. Gouze, Postnatal Soluble FGFR3 Therapy Rescues Achondroplasia Symptoms and Restores Bone Growth in Mice. Sci. Transl. Med. 5, 203ra124 (2013) DOI: 10.1126/scitranslmed.3006247
Abstract
Achondroplasia is a rare genetic disease characterized by abnormal bone development, resulting in short stature. It is caused by a single point mutation in the gene coding for fibroblast growth factor receptor 3 (FGFR3), which leads to prolonged activation upon ligand binding. To prevent excessive intracellular signaling and rescue the symptoms of achondroplasia, we have developed a recombinant protein therapeutic approach using a soluble form of human FGFR3 (sFGFR3), which acts as a decoy receptor and prevents FGF from binding to mutant FGFR3. sFGFR3 was injected subcutaneously to newborn Fgfr3ach/+ mice—the mouse model of achondroplasia—twice per week throughout the growth period during 3 weeks. Effective maturation of growth plate chondrocytes was restored in bones of treated mice, with a dose-dependent enhancement of skeletal growth in Fgfr3ach/+ mice. This resulted in normal stature and a significant decrease in mortality and associated complications, without any evidence of toxicity. These results describe a new approach for restoring bone growth and suggest that sFGFR3 could be a potential therapy for children with achondroplasia and related disorders.
© 2013 Medical Xpress