Bacteria-eating viruses 'magic bullets in the war on superbugs'
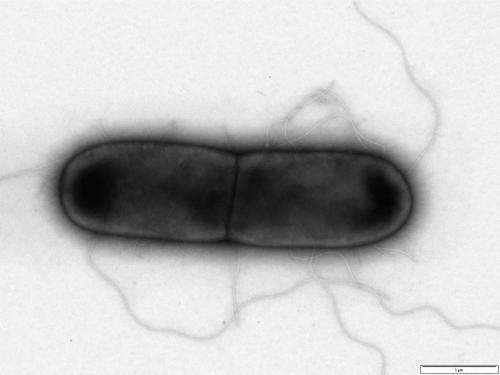
A specialist team of scientists from the University of Leicester has isolated viruses that eat bacteria—called phages—to specifically target the highly infectious hospital superbug Clostridium difficile (C. diff).
Now an exciting new collaboration between the University of Leicester, the University of Glasgow and AmpliPhi Biosciences Corporation could lead to the use of bacteriophages for treating the superbug Clostridium difficile infections.
Dr Martha Clokie, from the University of Leicester's Department of Infection, Immunity and Inflammation has been investigating an alternative approach to antibiotics, which utilizes naturally occurring viruses called bacteriophages, meaning 'eaters of bacteria'.
The work has predominantly been funded by the Medical Research Council (MRC).
Dr Clokie said: "Ever since the discovery of the first antibiotic, penicillin, antibiotics have been heralded as the 'silver bullets' of medicine. They have saved countless lives and impacted on the well-being of humanity.
"But less than a century following their discovery, the future impact of antibiotics is dwindling at a pace that no one anticipated, with more and more bacteria out-smarting and 'out-evolving' these miracle drugs. This has re-energised the search for new treatments.
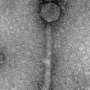
"One alternative to antibiotics is bacteriophages, known as phages, which unlike antibiotics, are specific in what they kill and will generally only infect one particular species, or even strain, of bacteria - referred to as the 'host'. Following attachment to their hosts, they inject their DNA into the bacterium, which then replicates many times over, ultimately causing the bacterial cell to burst open. The phages released from the dead bacterium can then infect other host cells."
Dr. Clokie and her team have achieved the remarkable feat of isolating and characterising the largest known set of distinct C. diff phages that infect clinically relevant strains of C. diff. Of these, a specific mixture of phages have been proved, through extensive laboratory testing, to be effective against 90% of the most clinically relevant C. diff strains currently seen in the U.K.
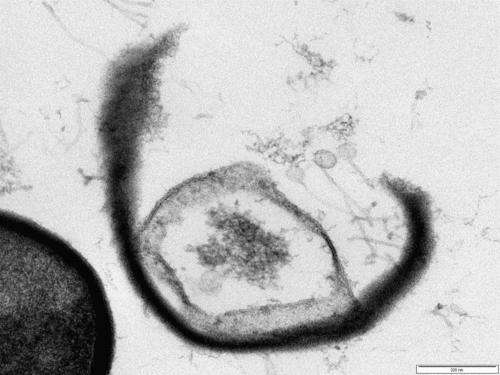
As a testament to their therapeutic potential, these phages, that are the subject of a patent application, have been licensed by AmpliPhi Biosciences Corporation - a US-based biopharmaceutical company and pioneers in developing phage-based therapeutics. AmpliPhi have already made progress in developing phages targeted against Pseudomonas aeruginosa, a pathogen that causes acute, life-threatening lung infections in cystic fibrosis patients. They were also the first biopharmaceutical company to demonstrate the effectiveness of Pseudomonas phages in controlled and regulated human clinical trials.
As part of this exciting collaboration, AmpliPhi are funding further development and testing of C. diff phages developed by Dr. Clokie. The goal is to have a phage mixture ready to go into phase 1 and 2 clinical trials. This will involve optimising phage-preparations for maximum efficacy against C. diff infections from around the globe and establishing production, storage and delivery systems for the phage mixture. Evaluations of the efficacy of bacteriophage therapy and optimisation of dosing regimes will be carried out in collaboration with the University of Glasgow, in the laboratory of Dr Gill Douce.
Dr Clokie said: "C. diff bacteria primarily affect our digestive system. Whilst relatively innocuous in individuals with a healthy gut flora, they pose a serious threat when our natural digestive environment is disrupted or depleted, such as after chronic antibiotic use.
In such individuals, C. diff infections can cause severe diarrhoea, vomiting and dehydration. Collectively, these symptoms can prove life threatening, particularly in elderly patients.
"In addition to killing the C. diff bacteria, antibiotics also destroy the 'good' gut bacteria, in turn increasing the potential for relapse or new infections. Consequently, C. diff infections pose a substantial healthcare burden for the NHS and a significant drain on its resources.
"The key advantage of using phages over antibiotics lies in their specificity. A phage will infect and kill only a specific strain/species of bacteria. This is particularly important when treating conditions like C. diff infections, where maintenance of the natural balance of gut bacteria greatly reduces the chance of relapse."
Phil Young, CEO and President of AmpliPhi, said "We are very excited about this partnership with Dr. Clokie and the University of Leicester. Phage-based therapy has the potential to revolutionise the way C. diff infections are treated in the clinic, in compliance with the regulatory frameworks already in place. We firmly believe that this collaboration may result in a treatment that could benefit patients, clinicians and health-care organisations alike."
Dr Des Walsh, Head of Infections and Immunity at the Medical Research Council, said: "Antibacterial resistance is a major and growing threat to health globally. New treatments and therapies are sorely needed. This study by Dr Clokie examines a new way to kill bacteria to circumvent resistance formation. She has established an impressive collection of "phage" viruses and has developed strong partnerships to translate her research into potential new treatments for Clostridium difficile infection—an excellent example of moving basic experimental MRC funded research along the development pipeline."
This collaboration also marks an important achievement for the University of Leicester and underlines its commitment to support cutting-edge scientific research that has the potential to benefit society at large.
Dr. Julie Pratt from the University Enterprise & Business Development Office, who led the negotiations with AmpliPhi, said: "This is a great example of how excellent scientific research can generate real business opportunities and promises to deliver a life changing impact on patients. It also reflects the growing strength of Leicester in the strategically important area of tackling the next generation of infectious diseases."
For Dr. Clokie, who has spent 6 years working on C. diff phages, these are very exciting times. "We hope our partnership with AmpliPhi will lead to the introduction of a much-needed change in the way C. diff infections are treated", she said.
"The use of phage-based therapy could eliminate the negative impact of antibiotics on the gut flora, minimise chances of relapse, provide patients with an effective, lasting safeguard against these life-threatening bacteria and relieve a substantial portion of the health-care and financial burden this superbug poses on the NHS. Ultimately, I hope this will pave the way for a greater use of bacteriophages in the wider, global fight against antibiotic-resistant bacteria."
Professor Mark Wilcox, Professor of Medical Microbiology, University of Leeds, and Lead on C. difficile for Public Health England, said: "New options to treat C. difficile infection (CDI) would be welcome, particularly if these do not damage the normal, friendly gut bacteria. Phage based therapy is an interesting possibility that should protect these helpful bacteria, which are important in patients with CDI."