New discovery could dramatically reduce leishmaniasis treatment doses and side effects
An international team of scientists, with the participation of the Universitat Autònoma de Barcelona, have developed a compound to treat leishmaniasis in humans using today's most commonly prescribed drug, but with an 83 percent increase in its effectiveness. The complex compound, a combination of the drug and nanoparticles which transport it to the infected cells, has been successfully tested with animal models of the disease. The research has been published in the Journal of Infectious Diseases.
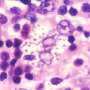
The Amphotericin B (AmB) is the main active ingredient in the most effective drug used to treat leishmaniasis, a disease which in the Western world mainly affects dogs, but in developing countries affects over 12 million people, with more than 70,000 deaths per year. The cost of treating humans with AmB surpasses $5,000 per patient, the treatment is extensive (2-hour sessions every day during 21 days), and the side effects are frequent and many times patients must be hospitalised.
Researchers from the University of Miami, Florida and the Universitat Autònoma de Barcelona have developed a method which allows to drastically reduce the drug dose, since it improves its efficacy 83%, multiplies by 10 the capacity of the drug to attack cell infected by the parasite which provokes the disease, and significantly reduces the toxicity of the parasite. The method has been successfully tested on mice models of leishmaniasis.
The complex compound acts through the action of the PDD nanoparticle, a substance measuring some 10 nanometres in diameter which is fit into the active principle, Amphotericin B, and guides it selectively to the cells harbouring the parasite. Scientists observed how, while the usual complete dose of the drug requires over 12 days to reduce the skin lesions caused by the disease, one dose of the complex compound with only 17% of the complete dose of the drug improves skin lesions in two or three days. Moreover, the complex compound acts as a therapeutic vaccine which activates the immune system against the reservoir cells hosting the parasite.
The PDD substance has been used in previous trials with people with the aim of improving the response of their immune system to other diseases. Now there is a need for clinical trials with humans in order to verify its safety as an adjuvant in the treatment of leishmaniasis. If its safety in humans is confirmed, it will also reduce the cost of the treatment drastically, and this is a key element in reducing mortality rates in developing countries.
The research, recently published in the Journal of Infectious Diseases, included the participation of lecturer Jordi Alberola and Dr Alhelí Rodríguez, from the Department of Pharmacology, Therapeutics and Toxicology and the Drug Analysis Service of the UAB.