November 7, 2013 report
The glial menagerie: From simple beginnings to staggering complexity
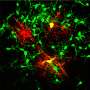
(Medical Xpress)—In preparation for neuroscience's huge SFN2013 meeting next week in San Diego, many key scientific journals are rolling out their special neuro-focus issues. Generally this is the time for the field to reflect, to dream, and for closed-access only journals, perhaps to cry. The current issue of Neuron has many fascinating reviews written by notable dignitaries in their area of expertise. The title above comes from a subsection of one of these reviews which is unique in that if offers a clear path to understanding something neuroscientists have struggled with to this day—namely, the function of glial cells, and from whence they came. The answer to this conundrum (and for myelinating cells in particular), is written play-by-play, often in freeze-frame, and sometimes even with rewind, in the structures of creatures not normally associated with having a significant myelin endowment. But these creatures, known to us generally as invertebrates, generate a huge variety of glial ensheathing cells. As we shall see, they run the entire gamut from simple shield-like elements, to full-blown spiralling membrane multi-wraps.
Neuroscientists talk a lot about the functions of the 302, or more, neurons found in C. elegans. This worm also has some 50 glial cells, which like neurons, come from ectodermal precursor tissues. It additionally has six glial sheath cells that, notably, hail from mesodermal origins. Four of these sheath cells in the cephalic area closely associate with various dendritic elements of key neurons in the worm. What is of interest here, is that these cells also extend a sheetlike investment to the nerve ring brain of the worm, which may be interpreted perhaps as the primitive form of a blood-brain-barrier.
Other invertebrates like Drosophila, still lack complex myelin per say, but coat their tracheal gas tubes and hemolymph-filled vasculature with elaborate glial process. Researchers have identified the "Glial Cells Missing" (GCM) gene in Drosophila as a critical regulator in these processes. Unfortunately for glial cell science, GCM orthologs in mammals apparently lack similar power and many comparative studies in this area have faltered. According to the authors, this situation prompted one prominent Stanford neurobiologist to lament, "Maybe fly glia are from outer space"?
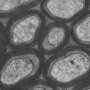
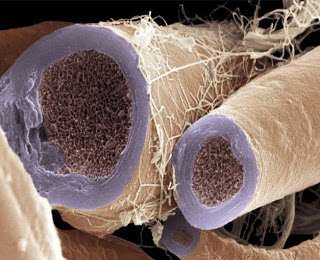
Fortunately there are other invertebrates which have managed more than rudimentary attempts to myelinate their connections. Their different design philosophies can be seen well in cross section where unique node geometries are immediately evident. Vertebrate myelin typically has a circumferential node placement, a feature thus far not found in invertebrates—unless one includes the Caridean shrimp. Earthworms (which can have up to 200 spiral myelin layers) as well as copepods, have what are known as "fenestrated" or "focal" nodes, which expose only a few small windows of axon to the exterior space.
In looking to better explain the rise of myelination, the authors ask a simple question, how do glial cells,like oligodendrocytes, fund their massive membrane production, and where do they get the building blocks? If neurons, with their long, high aspect processes are the classic example of cells gone wild into the linear dimension, then oligodendrocytes must be the same but for massive hypertrophy via surface area. It would seem that their 6500-fold foray into membrane production, which often occurs within a relatively short time, would require close associations with a vascular source, and a lipid-optimized version of the stand metabolic kit. Researchers have found that lactate is a critic source of energy for many varieties of glial cell, especially during myelination. Later on, glial-derived lactate can be in turn oxidized by neurons. Within axons themselves, various other sources of energy, aside from that supplied by mitochondria can be utilized.
The role of mitochondria in oligodendrocytes has not been a major concern to date in neurobiology. These subcellular creatures are no where near as abundant here as they are within the synaptic environment. However oxidation, or rather oxygen in general, may have been an important creative force in the evolution of myelination. At least in mammalian brains, it is interesting to note that myelination is basically a post-natal affair. Compared to the in-utero environment, we might expect the greater oxygen tension available to the devlopee could have some transformative effects, not least being myelination.
While plenty of idiosyncratic invertebrate snapshots of myelination already exist, it is perhaps only with more detailed feature films that lofty questions of how axon are actually wrapped by glia—and therefore how this white matter tag-team persists in flux with our longer term cerebral aspirations and memories—will be answered. The riveting particulars of various lipid phase transitions in myelin, and the life-long inexorable battles for directional and orientational supremacy of competing oliodendrocytic arms await our discovery.
The Neuron review only touches on a few of the major types of glial cells. Other varieties like the microglial cells, the immuno-agents of the brain, contain their own dynamic world unto themselves. The huge variety of glial cells in mammals seems to have paralleled a similar refinement in neuron types that have been expanded to fill every chemical niche of the hydraulo-metabolic net that is the brain. While only a small percent of the cells in the CNS of invertebrates are counted as glial cells, their progression into numerical dominance has continued unabated as complex brains evolve.
More information: The Neuron Identity Problem: Form Meets Function, Neuron, Volume 80, Issue 3, 602-612, 30 October 2013 www.cell.com/neuron/abstract/S0896-6273(13)00987-2
Abstract
A complete understanding of nervous system function cannot be achieved without the identification of its component cell types. In this Perspective, we explore a series of related issues surrounding cell identity and how revolutionary methods for labeling and probing specific neuronal types have clarified this question. Specifically, we ask the following questions: what is the purpose of such diversity, how is it generated, how is it maintained, and, ultimately, how can one unambiguously identity one cell type from another? We suggest that each cell type can be defined by a unique and conserved molecular ground state that determines its capabilities. We believe that gaining an understanding of these molecular barcodes will advance our ability to explore brain function, enhance our understanding of the biochemical basis of CNS disorders, and aid in the development of novel therapeutic strategies.
© 2013 Phys.org