November 5, 2013 report
Wiring up the visual system requires precise temporal control of axon terminations
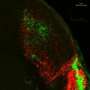
(Medical Xpress)—The Lateral Geniculate Nucleus (LGN) of the thalamus is a busy place, especially during development. Although it receives inputs from many regions of the brain, the first class seats are reserved for axons coming in from the retina. Brainstem activating projections, and feedback axons from the visual cortex, arrive at this thalamic terminal early on, but must wait until the retinal inputs have taken their seats before they can board. The molecular staff that coordinates this precise temporal arrangement has recently been identified, and the results publsihed in the journal Cell Reports. The authors show that the LGN is initially infused with aggrecan, a molecule that is particularly repulsive to cortico-thalamic axons. The aggrecan keeps them at bay for up to two weeks while the retinal axons get sorted. At that point, aggrecanase, which chops up aggrecan, is expressed in the LGN, and the thalamocortical axons can enter.
One question raised by the above, is if aggrecan is such a potent axon inhibitor, how do the retinal axons ever make it in to the LGN? There may be a few possibilities here, but one thing the authors note is that retinal axons express integrin receptors while cortical axons do not. Integrin receptors provide a way for cells to attach to surrounding extracellular matrix, or to other cells, and transduce regulatory signals to the interior of the cell. The more pressing issue, at least in this study, is how the expression of aggrecanase is regulated.
The authors suspected that it is the retinal axons themselves which control the timing of aggrecan degredation. They had previously shown that if the retinal inputs are removed in mice, either surgically or genetically, results, the cortico-thalamic axons enter the LGN sooner. When the researchers looked at the aggrecan protein levels in these animals, they found that the levels were greatly reduced, but that the mRNA expression levels were not. Based on further studies of mutant animals they propose that retinal inputs initially prevent expression of endogenous aggrecanases in the LGN. After they enervate the LGN, this brake is released, resulting in aggrecan degradation and subsequent enervation by cortical projections.
The authors note that the integrin effect alone may not be the full story of how the retinal axons manage to get a pass for the LGN. They note that retinal cells express receptors for various neurotrophins including neurotrophin 3 and BDNF. Both of these factors have been shown to be enriched in the LGN during the time period when enervation is at its peak.
Further studies might reveal a role for aggrecan distribution, and its regulation by retinal inputs, in the formation of specific patterns in the LGN. Pattern formation in the visual system has been studied extensively, and is known to be regulated by spiking activity in the input neurons. The precise molecular pathways by which this activity is converted into structure are still largely unknown, but future work could lead to exciting new developments in this area.
More information: A Molecular Mechanism Regulating the Timing of Corticogeniculate Innervation, Cell Reports, 31 October 2013. DOI: 10.1016/j.celrep.2013.09.041
Abstract
Neural circuit formation demands precise timing of innervation by different classes of axons. However, the mechanisms underlying such activity remain largely unknown. In the dorsal lateral geniculate nucleus (dLGN), axons from the retina and visual cortex innervate thalamic relay neurons in a highly coordinated manner, with those from the cortex arriving well after those from retina. The differential timing of retino- and corticogeniculate innervation is not a coincidence but is orchestrated by retinal inputs. Here, we identified a chondroitin sulfate proteoglycan (CSPG) that regulates the timing of corticogeniculate innervation. Aggrecan, a repulsive CSPG, is enriched in neonatal dLGN and inhibits cortical axons from prematurely entering the dLGN. Postnatal loss of aggrecan from dLGN coincides with upregulation of aggrecanase expression in the dLGN and corticogeniculate innervation and, it is important to note, is regulated by retinal inputs. Taken together, these studies reveal a molecular mechanism through which one class of axons coordinates the temporal targeting of another class of axons.
© 2013 Medical Xpress