HIV-1 movement across genital tract cells surprisingly enhanced by usurping antibody response
Infectious disease researchers have identified a novel mechanism wherein HIV-1 may facilitate its own transmission by usurping the antibody response directed against itself. These results have important implications for HIV vaccine development and for understanding the earliest events in HIV transmission.
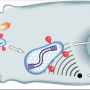
In a study appearing in the November issue of PLoS Pathogens, Dr. Donald Forthal of UC Irvine and colleagues studied the mechanisms employed by the virus to cross genital tract tissue and establish infection. Since cervicovaginal fluid is acidic and HIV-1 in cervicovaginal fluid is likely coated with antibodies, they explored the effect of low pH and HIV-1-specific antibodies on transcytosis, the movement of HIV-1 across tight-junctioned epithelial cells.
The researchers found that the combination of HIV-1-specific antibodies and low pH enhanced transcytosis as much as 20-fold.
Virus that underwent transcytosis under these conditions was infectious, and infectivity was highly influenced by whether or not the antibody neutralized the virus. They observed enhanced transcytosis using antibody from cervicovaginal and seminal fluids and using transmitted/founder strains of HIV-1. Enhanced transcytosis was due to the Fc neonatal receptor (FcRn), which binds immune complexes at acidic pH and releases them at neutral pH. Finally, staining of human tissue revealed abundant FcRn expression on columnar epithelial cells of penile urethra and endocervix.
More information: www.plospathogens.org/article/info%3Adoi%2F10.1371%2Fjournal.ppat.1003776