Beaumont Health System first outside of Japan to test new atrial fibrillation technology
Beaumont Health System is the first center outside of Japan to use a new balloon ablation technology to treat atrial fibrillation, the most common heart rhythm disorder that affects about 3 million people in the U.S.
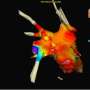
Named after its Japanese inventor, Dr. Shutaro Satake, the treatment uses a special catheter equipped with a balloon that is inflated with liquid then heated to burn or ablate the source of atrial fibrillation in the heart.
David Haines, M.D., director of Beaumont's Heart Rhythm Center, is the national principal investigator of the research study testing the safety and effectiveness of the device. The technology is not yet approved by the U.S. Food and Drug Administration for commercial use. It has been used in studies in Japan in more than 500 people. A total of 35 patients will take part in the study at five centers in the U.S.
"This treatment is designed for people with paroxysmal atrial fibrillation – a rapid, irregular heart rhythm that comes and goes on its own and has not responded to anti-arrythmic drugs," says Dr. Haines. "We are hopeful that this procedure will be safer, faster and as effective as current approved technologies for atrial fibrillation ablation."
People with atrial fibrillation have an irregular heartbeat that is sometimes too fast and causes symptoms like feelings of weakness, dizziness or lightheadedness and/or an uncomfortable racing or unusual heartbeat. The condition puts them at increased risk of stroke.
In the procedure, a small catheter, or hollow tube, is inserted through the femoral vein in the groin area and passed through the veins into the left atrial chamber of the heart. The Toray hot balloon is placed at the opening of the pulmonary vein – a large blood vessel that carries oxygenated blood from the lungs to the heart. The flexible balloon is inflated with liquid and a radiofrequency electrode within the balloon heats the liquid to burn or ablate tissue to block triggering impulses that cause atrial fibrillation.
Patients taking part in the study will return for follow-up visits up to 12 months after the procedure for routine cardiac testing to validate the results of the treatment. They will be given a trans-telephonic monitor to send their heart rhythm over the phone to a special center from discharge to six months after treatment.