Research team discovers molecule that may help fight cancer drug resistance

(Medical Xpress)—There are no popularity contests for proteins, but a prevalent protein called ubiquitin drew quite a following after three scientists received the 2004 Nobel Prize in Chemistry for discovering its role in mediating protein degradation.
That ground-breaking research on ubiquitination was conducted in the 1970s and '80s, and it has since been discovered that this small protein plays a big role in cell biology, including how the human body deals with internal and external attacks on its DNA.
However, while the essential roles of ubiquitin have received a lot of attention in the decade since the Nobel was awarded, the reverse of ubiquitination, or deubiquitination, is less well understood.
Now, a group of researchers led by Zhihao Zhuang, associate professor in the Department of Chemistry and Biochemistry at the University of Delaware, has discovered that a deubiquitinase (or DUB) complex, USP1-UAF1, may be a key regulator of the DNA damage response and a target for overcoming resistance to platinum-based anticancer drugs. Their findings were published in the online version of Nature Chemical Biology on Feb. 16.
Our DNA is under constant attack by UV light, radiation, industrial carcinogens, and other environmental factors, as well as by endogenous processes such as oxidation and hydrolysis. But our bodies also have ways to repair that damage or to tolerate it temporarily until a repair strategy kicks in.
Zhuang's research group is particularly interested in a DNA damage tolerance mechanism known as translesion synthesis (TLS), in which enzymes called TLS polymerases synthesize DNA over the damaged nucleotide bases, followed by normal replication after the lesion.

The problem is that while TLS polymerases are generally good guys, they also have a dark side—they have been implicated in making cancer cells resistant to certain cancer drugs, including cisplatin, which is often used to treat testicular, bladder, and ovarian cancers that have spread.
"Cancer drugs like cisplatin work by damaging DNA and thereby preventing cancer cells from replicating the genomic DNA and dividing," Zhuang explains. "However, cancer cells quickly develop resistance to cisplatin, and we and other researchers suspect that a polymerase known as Pol η is involved in overcoming cisplatin-induced lesions."
So while Pol η is a good guy under normal circumstances—in effect, paving the way over DNA potholes until the road levels out on the other side—it becomes a bad guy when the smooth road enables cancer cells to keep traveling.
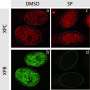
Now, Zhuang and his team have discovered a new molecule, ML323, that can inhibit processes, such as TLS, that may be hijacked by cancer cells to overcome the roadblocks on genomic DNA.
"Using ML323, we studied the cellular response to DNA damage and revealed new insights into the role of deubiquitination in both the TLS pathway and another one called the Fanconi anemia, or FA, pathway," Zhuang says. "We're very encouraged by the fact that a single molecule is effective at inhibiting the USP1-UAF1 DUB complex and disrupting two essential DNA damage tolerance pathways."
The finding may give clinicians a new weapon in the arsenal against cancer.
"We exposed lung cancer cells to cisplatin alone and to a combination of cisplatin and ML323, and we achieved close to an order of magnitude reduction in EC50, a common measure of the potency of small-molecule drugs, when the combination was used," Zhuang says. "This tells us that the inhibitor effectively restores the ability of cisplatin to fight cancer cells."
"Cancer treatment is increasingly moving to combination therapies to make it more effective and less toxic to normal cells," he adds. "I think our findings from this work indicate that the USP1-UAF1 DUB inhibitor not only shows promise in fighting cancer but also can help us in investigating the complex biology of DNA damage responses."
The Nature Chemical Biology paper has been selected as a highlight by several journals since its publication online last month, and many people have contacted Zhuang to express interest in the work.
More information: "A selective USP1–UAF1 inhibitor links deubiquitination to DNA damage responses." Qin Liang, et al. Nature Chemical Biology (2014) DOI: 10.1038/nchembio.1455 . Received 06 July 2013 Accepted 11 December 2013 Published online 16 February 2014