Researchers develop strategy to combat genetic ALS, FTD
A team of researchers at Mayo Clinic and The Scripps Research Institute in Florida have developed a new therapeutic strategy to combat the most common genetic risk factor for the neurodegenerative disorders amyotrophic lateral sclerosis (ALS or Lou Gehrig's disease) and frontotemporal dementia (FTD). In the Aug. 14 issue of Neuron, they also report discovery of a potential biomarker to track disease progression and the efficacy of therapies.
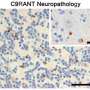
The scientists developed a small-molecule drug compound to prevent abnormal cellular processes caused by a mutation in the C9ORF72 gene. The findings come on the heels of previous discoveries by Mayo investigators that the C9ORF72 mutation produces an unusual repetitive genetic sequence that causes the buildup of abnormal RNA in brain cells and spinal cord.
While toxic protein clumps have long been implicated in neurodegeneration, this new strategy takes aim at abnormal RNA, which forms before toxic proteins in C9ORF72-related disorders (c9FTD/ALS). "Our study shows that toxic RNA produced in people with the c9FTD/ALS mutation is indeed a viable drug target," says the study's co-senior investigator, Leonard Petrucelli, Ph.D., a molecular neuroscientist at Mayo Clinic in Florida.
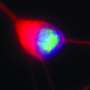
The compound, which was tested in cell culture models of c9FTD/ALS, bound to and blocked RNA's ability to interact with other key proteins, thereby preventing the formation of toxic RNA clumps and "c9RAN proteins" that results from a process called repeat-associated non-ATG (RAN) translation.
The researchers also discovered that c9RAN proteins produced by the abnormal RNA can be measured in the spinal fluid of ALS patients. They are now evaluating whether these proteins are also present in spinal fluid of patients diagnosed with FTD. Although ALS primarily affects motor neurons leading to impaired mobility, speech, swallowing, and respiratory function and FTD affects brain regions that support higher cognitive function, some patients have symptoms of both disorders.
"Development of a readily accessible biomarker for the c9FTD/ALS mutation may aid not only diagnosis of these disorders and allow for tracking disease course in patients, but it could provide a more direct way to evaluate the response to experimental treatments," says co-author Kevin Boylan, M.D., medical director of the Mayo Jacksonville ALS Center, the only ALS Certified Center of Excellence in Florida.
For example, a decrease in the levels of c9RAN proteins in response to treatment would suggest that a drug is having a desired effect. "The potential of this biomarker discovery is very exciting—even if we are in early days of development of such a test," he says.
Since ALS is usually fatal two to five years after diagnosis and there is currently no effective treatment for FTD, these landmark findings offer the possibility of both improved diagnosis and treatment for up to 40 percent of all patients with familial (inherited) ALS and up to 25 percent of patients with familial FTD, says Dr. Boylan.
"One of the most exciting aspects of these studies has, in my opinion, been the seamless collaboration of our Florida biosciences institutes—Scripps and Mayo. Our collective biological and chemical expertise made this research possible," says the other co-senior investigator, Mathew Disney, Ph.D., a professor of chemistry at Scripps Florida.
Dr. Disney and his group studied the structure of the RNA that resulted from the C9ORF72 mutation, and then designed the lead small-molecules. The Mayo team developed the patient-derived cell models to test the compounds in. Both teams then worked together to show that the lead agent's mode of action was targeting the toxic RNA.
More information: Neuron, Su et al.: "Biomarker and lead small molecule discovery to target r(GGGGCC)-associated defects in c9FTD/ALS." www.cell.com/neuron/abstract/S0896-6273(14)00673-4