September 1, 2014 report
Researchers find evidence of a link between high fat diets, gut bacteria, and bowel cancer
(Medical Xpress)—A large team of researchers, with members primarily from Germany, has found evidence that implicates high fat diets as a cause of bowel cancer due to a link with gut bacteria. In their paper published in the journal Nature, the team describes the results of research they conducted using mice that have a gene associated with the onset of bowel cancer.
Medical scientist have known for some time that there is a link between high fat diets and various cancers of the bowels, but until now were not able to explain why such a connection exists. Some have suggested that it's tied to obesity, or other side-effects that occur when a person eats a diet high in fat. In this new effort, the researchers have found evidence that exonerates obesity and instead implicates gut bacteria.
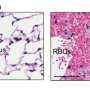
To learn more about the onset of bowel cancer, the researchers obtained mice with a genetic predisposition to the disease—they fed some of them on high fat diets and others on normal fat diets. They also took fecal samples to note the makeup of the microbial community in the gut, initially and then periodically thereafter. In so doing, they noted a shift in composition of the gut microbe composition over time in the mice on the high fat diet—there were more of some bugs, and less of others. The result they noted was a reduced immunity defense against bowel cancers with an associated increase in the rates of cancers observed.
To further bolster the evidence of the impact of gut biota in fighting off bowel cancer, the team killed off the biota community completely in some mice (with a predisposition to bowel cancer) and found the incidence of bowel cancers decreased. They also found that transferring samples of gut biota from predisposed mice that had bowel tumors to tumor-free (but still predisposed) mice—they too exhibited a higher rate of developing tumors than those that did not receive the transfer. These experiments, the researchers claim, indicate a very strong link between a high fat diet, gut biota and bowel cancer.
The research also suggests that in addition to modifying diet to reduce bowel cancers, there may be a way to lower the chances of getting bowel cancers by reestablishing normal gut biota in people who continue to eat diets high in fat.
More information: High-fat-diet-mediated dysbiosis promotes intestinal carcinogenesis independently of obesity, Nature (2014) DOI: 10.1038/nature13398
Abstract
Several features common to a Western lifestyle, including obesity and low levels of physical activity, are known risk factors for gastrointestinal cancers. There is substantial evidence suggesting that diet markedly affects the composition of the intestinal microbiota. Moreover, there is now unequivocal evidence linking dysbiosis to cancer development3. However, the mechanisms by which high-fat diet (HFD)-mediated changes in the microbial community affect the severity of tumorigenesis in the gut remain to be determined. Here we demonstrate that an HFD promotes tumour progression in the small intestine of genetically susceptible, K-rasG12Dint, mice independently of obesity. HFD consumption, in conjunction with K-ras mutation, mediated a shift in the composition of the gut microbiota, and this shift was associated with a decrease in Paneth-cell-mediated antimicrobial host defence that compromised dendritic cell recruitment and MHC class II molecule presentation in the gut-associated lymphoid tissues. When butyrate was administered to HFD-fed K-rasG12Dint mice, dendritic cell recruitment in the gut-associated lymphoid tissues was normalized, and tumour progression was attenuated. Importantly, deficiency in MYD88, a signalling adaptor for pattern recognition receptors and Toll-like receptors, blocked tumour progression. The transfer of faecal samples from HFD-fed mice with intestinal tumours to healthy adult K-rasG12Dint mice was sufficient to transmit disease in the absence of an HFD. Furthermore, treatment with antibiotics completely blocked HFD-induced tumour progression, suggesting that distinct shifts in the microbiota have a pivotal role in aggravating disease. Collectively, these data underscore the importance of the reciprocal interaction between host and environmental factors in selecting a microbiota that favours carcinogenesis, and they suggest that tumorigenesis is transmissible among genetically predisposed individuals.
© 2014 Medical Xpress