October 31, 2014 report
New viral tools for mapping brains
(Medical Xpress)—A brain-computer-interphase that is optogenetically-enabled is one of the most fantastic technologies we might envision today. It is likely that its full power could only be realized under the guidance of accurate maps of the brain's activity and connections. Creating these maps—or more likely, these dynamic models—will require tools that have scarcely been described let alone implemented. Among the most tantalizing ideas yet to emerge in this vein are probably what the "Kording-Church DNA tickertapes" which are based on modified DNA polymerases to record activity, and also "Zador neural barcodes" that are based on modified viruses which record connections.
When these two concepts were first described a few years ago, the initial excitement over them quickly gave way to the harsh realities of their complexity. Getting viruses to generate recombinant barcode labels for neuronal connections seems like pie in the sky when we can hardly get them to transport themselves to desired locations and express desired proteins without doing undue harm to cells. Tony Zador's group has now taken a big practical step towards contructing viral tools which use targeted gene expression to map neural circuits. Their latest work, published in Frontiers in Neuroanatomy, describes how they modified a pseudorabies virus (PRV) to efficiently navigate the nervous system and genetically tag select neurons.
Several kinds of viruses can invade the brain and turn it into their own private playground. Once past the blood brain barrier (BBB) , and safe from the normal immune sentinels that patrol the peripheral circulation, they bud across synapses within lipid bubbles and then hitch rides to more centralized hubs like winter enthusiasts on a retrograde axonal ski lift. The innate immune cells of the brain (the microglia), are generally powerless against viruses tucked away inside cells. In some cases of infection, drugs that permeabilize the BBB to let more capable immune agents enter the brain can be injected as a last resort, but the process is not very specific.
The PRV vector used by Zador's group has several advantages for mapping neurons compared compared to other potential viral vectors. One cousin of the PSV virus, namely a mutant of the HSV-1 virus, has been successfully used by others for retrogradely targeting particular neuronal populations. The main problem the HSV-1, is that like the brain-mapping molecular tickertapes, this guy is a proprietary and patented virus. Another flexible vector is a modified vesicular stomatitis virus (VSV-G). It can be readily switched between retrograde and anterograde transport modes with a change to a single gene. Unfortunately, many of the usual suspects that limit all things viral—namely infection inefficiency, cytotoxicity, limited payload capacity, safety concerns, availability, and unpredictable tropism—are all factors with VSV-G.
If you noticed we snuck the little word "tropism" in there. To really understand this technology we need to use at least a minimum of viral engineer-speak here. A virus with a penchant for neural tissue, like rabies, is said to show neurotropism. One that infects oligodendrocytes, like the JC virus, would have oligodendrotropism. Some of the magic used in the business is to take a viral particle with limited tissue expression, and change its specificity by "pseudotyping" with the coat proteins from another virus. Pseudotyping with rabies virus, for example, would give neural tissue compatability while for the above-mentioned VSV-G, pseudotyping with the glycoprotein G coat protein gives the ability to infect all tissue types.
As far as the PSV virus used by Zador's group goes, we need to put the brakes on the terminology train. The pseudorabies virus is not at all a pseudotyped rabies virus engineed for neurotropism. The pseudo simply comes from the fact that PSV in rabbits causes similar symptomes as rabies. In our present time of heightened viral awareness, the inherent species specificity, or lack thereof, goes a long way. PSV is not a human pathogen, and therefore it is not deadly to us like rabies virus is. However, if it is somehow introduced directly to neurons it will kill them in short order. The researchers were able to make PSV safe for neurons by blocking its IE180 gene, the single immediate early gene that acts as the master switch for the PSV replication cycle.
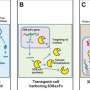
Without IE180 there is no PRV cytotoxicity, virus production, or spread across synapses. Unfortunately the PRV still needs to have a bit of that IE180 to engage in exclusive viral activities like retrograde transport. The trick Zador's group used was to modify the neurons themselves so that they can make the IE180 for the virus. That way there is more room to control not only how much is made, but which neurons can make it. The other major thing they did was to splice various flourescent proteins into the gaps of the reduced virus so that infected neurons could be tracked. This required adding something known as a site-specific Cre-loxP recombinase to control the fluorescent protein expression. (Cre comes from "Causes Recombination" while the recombination site itself, loxP refers to the "locus of crossing, P1).
The ultimate demonstration of neural omnipotence was to then apply the IE180-null PRV mutants in the expression of ligand or light-gated ion channels in neurons according to their specific projection targets. The optogenetic technique known as PINP (Photostimulation-assisted Identification of Neuronal Populations) could then be used to activate particular circuits and record their activity electrophysiologically. For this the researchers used the modified PRV virus to induce the expression of an optogenetically-active channel protein called ChR2 from yet another viral vector. In this case the second vector was an adeno-associated virus that they injected into the left auditory cortex of mice.
Now for the coup de grace; when the PRV was injected into the right-side cortex of these lucky mice, the stimulated fluorescence in the left cortex was confined to the specific 3-5 layer neurons which projected over to the right side. These neurons could be extracellularly recorded in vivo in response to brief flashes of blue light. This experimental state of the art is still a long way from molecularly-recorded records of activity or connections. None the less it is profound for its exacting use of the instruments that nature has already provided. The coordinated deployment of multiple customized viruses is proving to be the methodology of choice for navigating the brain on the small scales, and gaining the VIP access necessary to control it.
More information: Long-term Cre-mediated retrograde tagging of neurons using a novel recombinant pseudorabies virus, Front. Neuroanat., 03 September 2014, journal.frontiersin.org/Journa … nana.2014.00086/full
© 2014 Medical Xpress