Alirocumab shows promise as treatment to reduce LDL-cholesterol in Phase III study
A recently published clinical trial report reviewing the first completed Phase III study in the ODYSSEY development program has shown that alirocumab showed significantly better LDL-C lowering than ezetimibe, with a comparable safety profile to ezetimibe.
The report, which is published in the January issue of Future Cardiology, provides a comprehensive overview of the ODYSSEY MONO trial, including detail on the study design, data analysis, results and discussion on the implications of the findings, authored by lead investigator of the trial Eli M Roth.
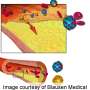
LDL-cholesterol (LDL-C) is considered to be a major modifiable risk factor for the development of atherosclerosis and cardiovascular disease (CVD), the leading cause of death worldwide. LDL-C is identified as the primary target of cholesterol-lowering therapy in both North American and European guidelines. Statins are the recommended first-line therapy for lowering LDL-C.
Alirocumab (formerly SAR236553/REGN727) is a fully human mAb to PCSK9 - the first in this class of drugs to complete a Phase III trial, and reported to have a significant role in the regulation of LDL-C - being developed jointly by Sanofi (France) and Regeneron (NY, USA).
This first completed Phase III study, entitled ODYSSEY MONO, tested the new lower 75-mg dose of alirocumab subcutaneously every 2 weeks as a monotherapy versus ezetimibe 10 mg per os every day as a control. Inclusion criteria included patients with an LDL-C between 100 mg/dl (?2.6 mmol/l) and 190 mg/dl (<4.9 mmol/l; inclusive) and not on LLT. In addition, patients needed to have moderate CV risk defined as a 10-year risk of fatal CV events ?1% and <5% based on the European Systematic Coronary Risk Estimation (SCORE).
These data from the study suggest that the 75-mg dose of alirocumab subcutaneously every 2 weeks may be appropriate for adequate lowering of LDL-C in a large portion of patients with primary hypercholesterolemia at moderate CV risk who are not receiving statin therapy.
The safety parameters, adverse events and study discontinuation rates were similar between both treatment arms.
"Since ODYSSEY MONO, we have seen several phase III trials that have confirmed the ability of alirocumab to lower LDL-C in moderate to high risk cardiovascular patients," said Eli M. Roth Medical Director, Sterling Research Group & Professor of Clinical Medicine University of Cincinnati, and lead author of the article published in Future Cardiology. "These completed trials show that PCSK9 inhibitors are proving to be effective in different patient populations."
Sean Fitzpatrick, Commissioning Editor of Future Cardiology, commented: "Following the presentation of Sanofi/Regeneron's positive top-line results at the end of 2014, we feel it is important for our readers to have a comprehensive, accessible overview of this important topic. The review article helps in providing context for clinicians and researchers framing the results in terms of the current state of the art in lipid-lowering therapy."
More information: Roth EM, McKenney JM. ODYSSEY MONO: effect of alirocumab 75 mg subcutaneously every 2 weeks as monotherapy versus ezetimibe over 24 weeks. Future Cardiology, 2015; 11(1): 27-37