March 30, 2015 report
Researchers target two brain sites responsible for anxiety
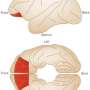
(MedicalXpress)—As distinct from a fear response to a real, immediate threat, anxiety is an expectation of future threat. The symptoms of anxiety disorders and the treatment of those symptoms are largely understood, but the neurology of anxiety is challenging to study. For instance, researchers are aware that dysregulation of the orbitofrontal and ventrolateral prefrontal cortices are implicated in anxiety and mood disorders, but the specific contributions of each region are not known.
A group of researchers at the University of Cambridge designed a study to assess the contributions of these regions in the anxiety responses of marmosets. They have published their results in the Proceedings of the National Academy of Sciences.
Overreaction to threat conditions
In patients with anxiety, the fear of negative outcomes has a strong negative influence on decision making, often leading to distress, social isolation and adverse health conditions. Sensitivity to threat is critical for the survival of an organism, but hypersensitivity and overestimation of future threats inhibit proper cost-benefit decision making in patients with anxiety and mood disorders.
Previous studies have suggested that this hypersensitivity is due to dysregulation within the prefrontal cortex, but questions persist regarding how this region affects aversive processing and the impact of negative emotional valence in decision making.
Knowing that excitotoxic lesions on either the anterior orbitofrontal cortex or ventrolateral prefrontal cortex heighten anxiety and fear responses in marmosets, the researchers designed an experiment to study the responses of test monkeys with temporary inactivation of these regions.
Testing the brain's threat prediction ability
The marmosets were trained to respond to two identical visual stimuli presented on each side of a touchscreen to gain a reward of banana juice. The visual stimuli were presented on independent but identical variable-interval schedules so that the optimal strategy for maximizing reward delivery would be a relatively equal response to both stimuli.
In the course of the experiment, researchers observed the response biases of individual monkeys; each monkey demonstrated a slight bias for one side of the screen or the other. Once per week, responses on one of the two stimuli would produce delivery of a punishment: an aversive loud noise that was superimposed on the unchanged reward schedule. In order to avoid spatial bias contributing to a punishment-induced bias, the punishment was always introduced on the individual monkey's "preferred" side.
In the absence of reward, the sound produced a strong aversive response. However, when the punishment and reward were delivered simultaneously, the monkeys did not alter their behavior; the researchers conclude that the banana juice reward was "worth" responding for, despite the possibility of the punishment.
Shutting down the cortices
The researchers then conducted the same experiment after the inactivation of either the anterior orbitofrontal cortex or ventrolateral prefrontal cortex with a GABA agonist 20 minutes before test sessions. The inactivation of one of the two regions had no effect on responses when the test produced reward but no punishment. However, when punishment was introduced for responses on one side of the screen, animals with inactivation of the ventrolateral interior prefrontal cortex produced a strong, immediate response bias away from the punishment.
The researchers observed no delayed or long-lasting effects when those animals were tested the subsequent day with a reward-only session. Bias developed in test animals during the reward-punishment sessions, but did not endure. Control animals that were infused with saline instead of the GABA agonist showed unchanged responses to the test in the presence of both reward and punishment.
However, animals with inactivated anterior orbitofrontal cortices showed no effects during reward-punishment sessions, but did show a profound bias away from the previously punished side the next day. Thus, the researchers associate the activity of the vlPFC with cost-benefit analyses and the antOFC with the consolidation of memory for the punishment.
The amygdala-hypocampal circuit
Theorizing that the formation of memory in antipunishment bias was based in the amygdala and hippocampus of the marmosets, the researchers cannulated the amygdalas and anterior hippocampi of the antOFC-cannulated animals. They found that the inactivation of either structure eliminated the antipunishment bias in those animals, confirming those structures as agents for the consolidation of punishment memory.
Disconnecting the amygdala from the hippocampus had the same effect as bilateral inactivations of either structure, indicating that punishment memory formation is subserved by the amygdala-hippocampal circuit.
The researchers suggest that in the future, cognitive behavioral therapy for patients afflicted with anxiety disorders could be tailored to correspond with the patient's ability to make accurate cost-benefit analyses or to form memories that lead to strong antipunishment biases. Such a determination could also point to either the ventrolateral prefrontal cortex or the anterior orbitofrontal cortex as therapeutic targets.
More information: "Regional inactivations of primate ventral prefrontal cortex reveal two distinct mechanisms underlying negative bias in decision making." PNAS 2015 ; published ahead of print March 16, 2015, DOI: 10.1073/pnas.1422440112
Abstract
Dysregulation of the orbitofrontal and ventrolateral prefrontal cortices is implicated in anxiety and mood disorders, but the specific contributions of each region are unknown, including how they gate the impact of threat on decision making. To address this, the effects of GABAergic inactivation of these regions were studied in marmoset monkeys performing an instrumental approach–avoidance decision-making task that is sensitive to changes in anxiety. Inactivation of either region induced a negative bias away from punishment that could be ameliorated with anxiolytic treatment. However, whereas the effects of ventrolateral prefrontal cortex inactivation on punishment avoidance were seen immediately, those of orbitofrontal cortex inactivation were delayed and their expression was dependent upon an amygdala–anterior hippocampal circuit. We propose that these negative biases result from deficits in attentional control and punishment prediction, respectively, and that they provide the basis for understanding how distinct regional prefrontal dysregulation contributes to the heterogeneity of anxiety disorders with implications for cognitive-behavioral treatment strategies.
© 2015 MedicalXpress.com