Research explains the formation of long-term motor memory
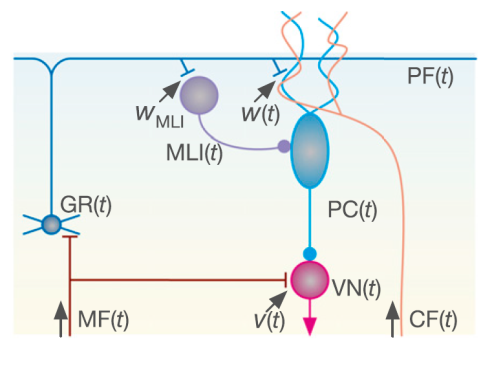
Recent studies of long-term motor memory have pointed out the involvement of synaptic plasticity at multiple sites in the cerebellum, but the physiological mechanism remains unclear. Now results from a collaboration of researchers at the University of Electro-Communications and the RIKEN Brain Science Institute in Japan, and the University of California, San Diego, in the US, successfully integrated the multiple plasticity mechanisms to explain the formation of long-term motor memory using simulations based on a mathematical model.
Until recently motor memory was widely considered the result of 'long-term depression' - a state of reduced efficacy following a stimulus - in the synapses at output neurons in the cerebellum called Purkinje cells. Yamazaki and colleagues developed a model for the optokinetic response (OKR) in eye movement that incorporated long-term potentiation - a state of increased efficacy following a stimulus - in the synapses at vestibular nuclear neurons. Thus, the model incorporates two distinct plasticity sites that function synergistically.
The model accurately reproduced experimental results. It showed that an hour's training resulted in a short-term increase of OKR gain, for which long-term depression at the Purkinje cells are responsible. Repetition of this training once a day gradually increased the level of OKR gain after training rather than during it, for which long-term potentiation at the vestibular nucleus neurons are responsible.
"It thus appears as if short-term memory formed in the Purkinje cells during 1-hour training is transferred to the vestibular nuclear neuron after training to consolidate as long-term memory," concluded the researchers. Their model also reproduced characteristics of the OKR behaviour observed in genetically manipulated mice.
More information: Yamazaki T, Nagao S, Lennon W and Tanaka S "Modeling memory consolidation during posttraining periods in cerebellovestibular learning." PNAS 112(11): 3541-3546 (2015). DOI: 10.1073/pnas.1413798112