Research finds key to gene stability, possible new cancer targets
Researchers in the UC Department of Cancer Biology have found new protein players that help keep genome stability—a promising finding with the potential to help with the development of new targeted therapeutics in diseases with genetic instability, such as cancer.
These findings were published in the March 2, 2015, issue of the journal Proceedings of the National Academy of Sciences (PNAS). The lead author is research associate and postdoctoral fellow Chunmin Ge, PhD, who is in the lab of Chunying Du, PhD.
"DNA damage repair is crucial for keeping genes stable," says Du, who is also a member of the Cincinnati Cancer Center. "Unrepaired DNA strands, particularly DNA double strand breaks, are highly toxic to cells. If left unrepaired, DNA double strand breaks often result in gene mutations which enhance malignant cell growth and tumor development.
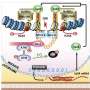
"In this study, we found a previously unknown function for an apoptotic inhibitor protein called BRUCE—apoptotic inhibitors hinder cell death—and a deubiquitinase called USP8, which controls a number of other metabolic functions in our body. They work together to promote DNA damage repair and prevent genetic instability. BRUCE and USP8 do their job by permitting the movement of a well-known tumor suppressor BRIT1 to the sites of DNA damage."
"We were also able to define the molecular pathway in which BRUCE and USP8 activate BRIT1 to maximize cell responsiveness to DNA damage," says Ge. "BRUCE holds the three proteins together to allow USP8 to remove the ubiquitin protein chains from BRIT1 to unleash BRIT1 to the DNA damage sites to fix DNA breaks."
Collaborators on this study include Lixiao Che, PhD, second author of the paper and postdoctoral fellow from Du's lab, as well as researchers from the Second People's Hospital of Lanzhou, China; the Houston Methodist Research Institute; Baylor College of Medicine; and Washington University.
"This work is very significant," says Jun-Lin Guan, PhD, Francis Brunning Chair and Professor in the Department of Cancer Biology. "It reveals the connections between two important cellular processes critical in cancer regulation and identifies the molecular mechanism involved. It is the accumulation of years of careful and painstaking studies of basic cancer mechanisms in the laboratory. This type of novel and creative basic research is crucial for fueling the future development of new cancer therapies."
Du says this study provides new insight into how removal of ubiquitin chains is important for the regulation of DNA repair and that her team will now work to identify mutations in this BRUCE-USP8-BRIT1 pathway that may link to development of diseases resulting from DNA repair defects and genome instability.
"We are pleased to have this collaborative work published," says Du. "Our findings provide important information to the rather complicated puzzle of the DNA damage response network."
More information: "BRUCE regulates DNA double-strand break response by promoting USP8 deubiquitination of BRIT1" PNAS 2015 ; published ahead of print March 2, 2015, DOI: 10.1073/pnas.1418335112