Can 'off the shelf' stem cell therapy treat heart failure patients? UB researcher aims to find out
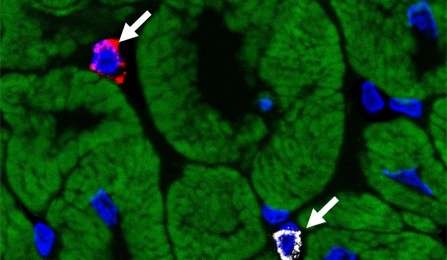
After a heart attack, cardiac stem cell therapy stimulates the growth of new heart cells, but exactly how that happens is unclear. The National Institutes of Health has awarded a $2 million grant to a researcher at the University at Buffalo School of Medicine and Biomedical Sciences to do a preclinical study to find out. The answer will help determine which type of cardiac stem cell therapy will be most effective in treating patients with heart failure.
Heart failure occurs when the heart cannot pump enough blood and oxygen to support the body's organs. According to the Centers for Disease Control and Prevention, it affects more than 5 million people in the U.S., with half of them dying within five years of diagnosis.
"We're trying to find out whether one of the cell types—either cardiosphere-derived cells (CDCs), which come from donors, or cardiac stem cells (CSCs) from the patient—are more effective when infused into the coronary arteries," says John M. Canty Jr., MD, Albert and Elizabeth Rekate Professor, chief of cardiology in the UB Department of Medicine and a cardiologist with UBMD.
CSCs reside in the heart, Canty explains, and some researchers hypothesize that they are the source of new cardiac muscle cells. They normally divide slowly, but when selectively harvested from a patient, they can be increased in number and then infused back into the recipient's heart. "After injection, the new cardiac stem cells proliferate and are the source of new heart muscle cells," says Canty. "With this approach, only cells from the same patient can be used."
Another approach is to take tissue from heart biopsies, which can grow into multicellular CDCs. Even though they do not develop into new muscle cells themselves, these cells can improve cardiac function. Canty and his colleagues previously demonstrated that these CDCs, as well as mesenchymal stem cells isolated from bone marrow, stimulate the recipient's own heart muscle cells to divide again. Canty notes this approach makes it potentially feasible to use stem cells from an unrelated donor and not the patient.
"Using cardiac stem cells that don't need to be harvested from the recipient would make this type of therapy more widely available to the increasing number of patients in need of treatments for heart failure," says Canty. "Demonstrating the feasibility of using cells from an unrelated donor would also provide an off-the-shelf approach to cardiac stem cell therapy."
He adds that infusing stem cell formulations directly into coronary arteries delivers the cells throughout the entire heart. This method is much simpler than injecting cells directly into heart muscle, which requires equipment that's not widely available.
If it turns out that harvesting a patient's own cells is more beneficial, then one approach could be to remove them with a minimally invasive heart biopsy, boost their numbers by culturing them outside of the body and then return them to the patient. While conceding that this method is time-consuming, Canty and his colleagues already have done this in preclinical studies. The method also is being used in clinical trials of other types of therapies.