Surgery followed by ipilimumab in melanoma patients improves survival
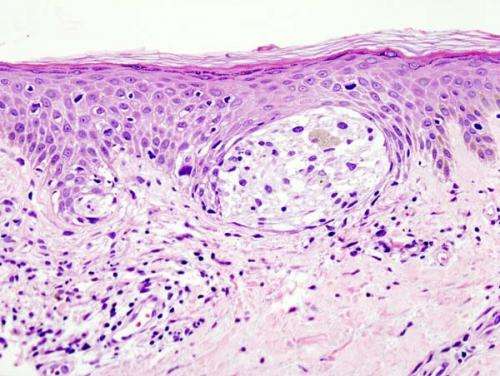
Melanoma is predicted to result in approximately 10,000 deaths in 2015. The majority of these deaths are due to advanced stage disease that has spread or metastasized to other sites. The prognosis for patients with metastatic melanoma remains poor, with 5-year survival rates of 63 percent in patients who have metastatic disease in regional lymph nodes, and only 17 percent in patients who have metastatic disease in distant sites. Moffitt Cancer Center researchers participated in an international phase 3 study that demonstrated that a drug called ipilimumab improves the relapse-free survival of advanced stage melanoma patients rendered free of disease surgically but at high risk for relapse.
Ipilimumab is approved for the treatment of melanoma that cannot be surgically removed or that has metastasized to different sites. Ipilimumab targets a protein called cytotoxic T lymphocytic antigen-4 (CTLA-4) that is found on a type of immune cell called a T cell. CTLA-4 keeps the immune system in check to avoid autoimmune disease by downregulating T cell activity. Tumors take advantage of CTLA-4 activity to avoid immune detection. By targeting CTLA-4, ipilimumab restimulates the immune system to target tumor cells.
Researchers wanted to determine if ipilimumab could improve the survival of advanced-stage melanoma patients if it was given after the surgical removal of both their primary melanoma tumors and their regional lymph nodes.
The study, which included researchers from 19 countries, compared ipilimumab treatment in 475 people to placebo treatment in 476 people. Patients who were administered ipilimumab survived longer without experiencing melanoma recurrence, with an average recurrence-free survival of 26.1 months in ipilimumab treated patients and 17.1 months in placebo treated patients. Additionally, 46.5 percent of ipilimumab treated patients survived 3 years without experiencing recurrence, while 34.8 percent of placebo treated patients survived 3 years without recurrence.
The researchers wanted to determine if particular patient characteristics were associated with a better response to therapy. They discovered that patients who had microscopic metastatic disease in their regional lymph nodes and a tumor that had a breakdown of the overlaying skin responded best to ipilimumab treatment.
Despite these promising results, ipilimumab treatment was associated with significantly higher toxicity than placebo treatment, including higher rates of gastrointestinal disorders, alterations in kidney function and inflammation of the pituitary gland. These adverse events resulted in 52 percent of patients on ipilimumab discontinuing treatment before its completion, including 39 percent of patients during the first four doses of ipilimumab. Furthermore, 5 patients on ipilimumab died during treatment due to adverse events.
The dose used in this study was significantly higher than the dose commonly used in melanoma patients. Additional studies are required to ensure that the benefit of this dose of ipilimumab does not outweigh the added toxicity risks.
The study was published on May issue of The Lancet. Funding support was provided by Bristol-Myers Squibb.
More information: The Lancet, www.thelancet.com/pdfs/journal … lanonc/PIIS1470-2045%2815%2970122-1.pdf