Re-booting the human gut
For decades, American travelers to international destinations have been plagued by acute gastrointestinal illnesses that can arise from travel to other countries. The Center for Disease Control (CDC) warns that depending on the destination, between 30 to 70 percent of travelers can expect to experience gastrointestinal distress from ingesting foreign or pathogenic bacteria that can be present in poorly sanitized water or food.
But novel biotechnology under development at Harvard's Wyss Institute for Biologically Inspired Engineering could help alleviate acute gastrointestinal illness for not only travelers abroad but also those who suffer from chronic gastrointestinal disorders, inflammation, or acute illnesses on home soil.
A new grant awarded by the Defense Advanced Research Projects Agency (DARPA), for up to $4.7 million dollars over the course of the work, will support the efforts of the project's co-principal investigators Wyss Core Faculty member Pamela Silver, Ph.D., and Wyss Senior Staff Scientist Jeffrey Way, Ph.D., who will team up with Wyss Founding Director Donald Ingber, M.D., Ph.D. The cross-disciplinary Wyss team aims to fight gastrointestinal illness through tactics invisible to the naked eye by developing an army of genetically engineered bacteria designed to sense, report and combat harmful microbial invaders.
"Travelers' illness is a huge problem for the military too," said Silver, who is also Professor in the Department of Systems Biology at Harvard Medical School. "There are lots of troops who are down for a period of time upon deployment overseas which has implications for safety and strategy- especially in the most severe cases when troops must be returned home for treatment."

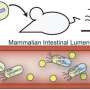
Silver and Way will hijack Nature's own mechanisms to create genetically engineered bacteria that are programmed to identify and kill harmful pathogenic bacteria. Building off their previous pioneering work, the synthetic bacteria will be engineered to detect the chemical signature given off by gastrointestinal inflammation. Then, once inflammation is detected, a series of genetic circuits in the synthetic bacteria will be triggered to attack invading bugs and restore healthy equilibrium in the gastrointestinal tract.
Taken in probiotic pill form, the microbial task force could potentially result in greatly reduced length of illness, returning military personnel and vacationers to their activities much sooner and with less chance of serious side effects.
Yet what enters the gastrointestinal tract will eventually exit - posing the challenge of keeping post-use probiotics from having unintended impact on the environment. Silver and Way will design their microbes to work collaboratively, employing a consortia of engineered bacteria that are only activated in a specific environment and when triggered with specific chemical signals - such as the kind of environment only found inside an inflamed human gastrointestinal tract.
"The consortia effect will basically act as a fail-safe," said Way. "It will block any unintended exchange of genetic information with species in the environment by preventing the probiotic consortia from being active and potent in any location outside of a human gut experiencing a specific kind of gastrointestinal inflammation."
To test and analyze the consortia in vitro through each stage of development, Ingber, who is also the Judah Folkman Professor of Vascular Biology at Harvard Medical School and Boston Children's Hospital and Professor of Bioengineering at the Harvard John A. Paulson School of Engineering and Applied Sciences, will employ the human gut-on-a-chip technology, which is one of several microfluidic devices making up the line of trailblazing human organs-on-chips technology that has emerged from his laboratory at the Wyss Institute.
The gut-on-a-chip is capable of sustaining living human gut cells in direct contact with the living microbiota within a microfluidic silicone chip for up to several weeks in vitro. Using the gut-on-a-chip, the team will mimic gastrointestinal inflammation with living human cells to study the pathogens' response to the collaborative consortia of engineered microbes. In addition, to prevent unintended environmental interactions, the gut-on-a-chip will be used to model the species-specific chemical signals that will trigger the consortia's exclusive activation.
The compelling work, which will be carried out over the next two and a half years, is now officially underway and also involves Georg Gerber, M.D., Ph.D., M.P.H., and Lynn Bry, M.D., Ph.D., from Brigham and Women's Hospital (BWH), who will carry out in vivo studies, and Daniel Gibson, Ph.D., of the J. Craig Venter Institute (JCVI), who is a world leader in the total synthesis of bacterial genomes. Working collaboratively, the group hopes to lay groundwork for understanding and influencing the human gut microbiome - the collective genome of the millions of micro-organisms that reside inside the body, which outnumber the human cells in our bodies by a ratio of ten to one.
"This multi-institutional effort to re-program the human gut microbiome not only has implications for treating gastrointestinal illnesses, but also opens doors to new ways of treating countless other diseases that are impacted by the microbiome, which represents a vast new frontier in medicine," said Ingber.