New device measures glucose in urine, ending annoying needle pricks

Two young technologists from the Technological Institute of Chihuahua in the north of Mexico have developed a device that can measure glucose in the blood by analyzing urine. It activates via smart phone bluetooth, recording daily values directly from the toilet.
Called Glucosalarm, it has achieved international recognition. Carlos Bernal, who created the device in collaboration with Nancy Guerra, said that considerations in the device design was the pain caused by the use of needles for monitoring glucose in blood, which is often why patients with diabetes do not maintain proper control of their disease.
"Pricking a finger several times a day can lead to numbness and over-sensitivity to the simple touch of clothing. Even the whole hand can become too painful to use for three or four days," says Bernal.
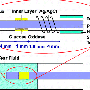
The device installed in the toilet. "The patient activates the sensor via bluetooth from a smartphone. Then, when urinating, a few drops are deposited on the collector and mixed with enzymes that react with the glucose and produce a colored compound; the sensor measures the intensity of the color, calculates the concentration of glucose, and sends the results to the phone in 15 to 40 seconds. If the result is too high, an alert is sent to the family, the doctor, and even to an emergency number requesting an ambulance.

"Using the smartphone, the patient can send the results to a doctor, family member or diabetes educator responsible for treatment, so that they are aware of the day-to-day glucose measurement," says Bernal.
Another reason for lapses in monitoring is the cost. "A glucometer costs less than 30 dollars, but the test strips are expensive, as are needles and needle holders, which is money that not everyone has. Glucosalarm will cost approximately $120 to $140, and the readings will cost less than a penny per measurement," says the engineer.
Currently, the device is in the third prototype and tests are performed with patients. It also has a patent in Mexico and the United States, and Carlos Bernal says two medical device companies with international presence have shown interest in manufacturing it.
Glucosalarm is expected to be released in the United States and Mexico in late 2016 and early 2017, once the necessary authorizations are obtained from the Food and Drug Administration (FDA) in the U.S. and the Federal Commission for the Protection against Sanitary Risk (COFEPRIS) in Mexico.