Novel insights in the attachment of the bacterial carcinogen Helicobacter pylori
The gastric pathogen Helicobacter pylori is highly adapted to survival in the human stomach and responsible for the majority of peptic ulcer and gastric cancer cases worldwide. An important survival strategy for the bacterium involves the tight binding to the stomach mucosa, out of reach of the noxious gastric juices. To do so, H. pylori adheres to blood group sugars found on gastric mucus and underlying cells.
Scientists at the labs of Han Remaut at VIB and Vrije Universiteit Brussel, and Thomas Borén at Umeå University in Sweden, provide detailed structural and functional insights into the protein responsible for this interaction. This BabA protein proves to be a molecular chameleon that adapts it binding properties and preference for different ABO-blood group sugars according to their prevalence in different human populations. However, the new study also uncovers BabA's Achilles heel: a short disulfide-bound loop that ropes around a critical sugar unit in the blood group antigens, an interaction that is broken by reducing pharmaceutics like N-acetylcysteine.
Snapshots of a molecular chameleon
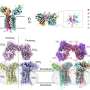
H. pylori has been associated with humankind since ancient migrations and is today still present in about halve of the world population. The bacterium has a fast evolving genome and is constantly adapting to changing conditions in its host. Kristof Moonens and coworkers now report in Cell Host & Microbe how the high sequence variation in the blood group antigen binding adhesin BabA leads to the functional polymorphism in its binding characteristics.
Kristof Moonens (VIB/VUB): 'By determining the X-ray structures of different BabA proteins, we could establish a general framework for ABO blood group binding by the adhesin. Thomas Borén's group had previously shown that "specialist" H. pylori strains only bind to gastric tissue of blood group O individuals, whereas "generalist" strains interact with all types of blood group individuals. Now we can show that a select network of residues in the protein steer the differences in binding preferences.'
Perspectives for H. pylori eradication therapy
Resistance of bacterial pathogens to antibiotics is becoming a widespread problem, and H. pylori proofs not different. Today, H. pylori eradication therapy already needs sustained treatment with a cocktail of 2-3 antibiotics. The quest for new treatment options or a vaccine is on, in particular in regions where H. pylori infections lead to a gastric cancer and ulcer epidemic. The new study provides perspectives to develop drugs that disrupt the bacterium's ability to hold on to the stomach mucosa.
Thomas Borén (Umeå University, Sweden): 'We could show that treatment with the redoxactive pharmaceutic N-acetylcysteine annihilates BabA function and furthermore that N-acetylcysteine lowers gastric mucosal neutrophil infiltration, i.e. stomach inflammation, in H. pylori-infected mice, providing perspectives on possible H. pylori eradication therapies. The additive effect of N-acetylcysteine on antibiotic eradication therapies has previously been reported in literature and now we have found a molecular basis and explanation for this effect.'
Han Remaut (VIB/VUB): 'Amidst BabA's receptor binding site that is constantly changing lies a conserved anchor point, a short disulfide-bound loop that embraces a fucose residue in the ABO blood group sugars. This newly elucidated structural element is inactivated by reduction and forms the basis for the rational design of novel anti-adhesive drugs that would reduce bacterial attachment, stomach inflammation and hence lower the risk for overt disease development.'
More information: Structural Insights into Polymorphic ABO Glycan Binding by Helicobacter pylori; Moonens et al. 2015, Cell Host & Microbe; DOI: dx.doi.org/10.1016/j.chom.2015.12.004