Pediatric sickle cell study stopped early due to positive results
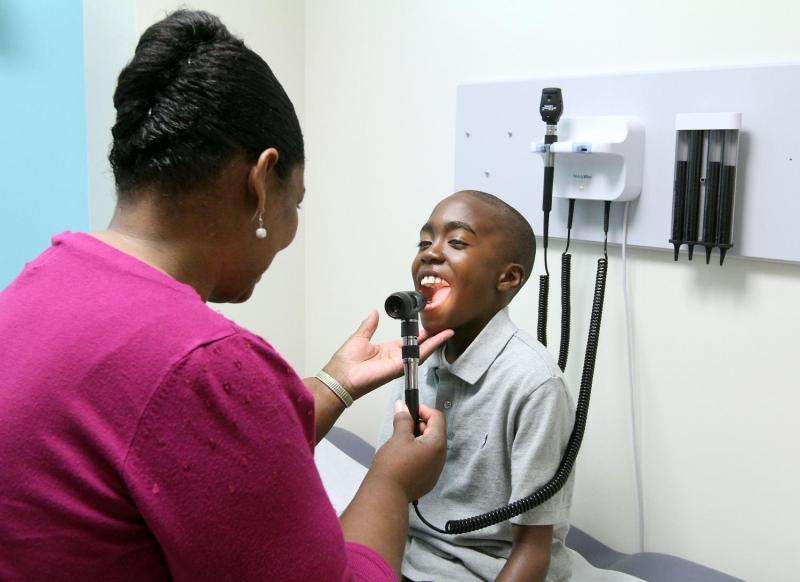
A national sickle cell disease study involving Medical University of South Carolina researchers found that for some children with sickle cell disease, the drug hydroxyurea is as effective as blood transfusions to reduce blood flow speeds in the brain. Increased blood flows are a major risk factor for stroke in these children. Study findings were published Dec. 6 in The Lancet and were presented at the American Society of Hematology meeting.
"It was a privilege to be a part of this well-designed and executed study. Russell Ware presented the results at the ASH meeting, and 18 years ago, almost to the day, I presented the STOP study results to the same meeting," said Robert J. Adams, M.D., study principal investigator, MUSC professor of neurosciences and director of the South Carolina Stroke Center of Economic Excellence. "That study showed how effective transcranial Doppler risk stratification, followed by regular red cell transfusions in those with high risk blood flow, can be in the prevention of stroke in these children. This became known as the STOP protocol and its wide adoption has been associated with a sharp drop in ischemic strokes in children with sickle cell disease. The drawback of indefinite transfusions however, was a limitation to wider use of the STOP protocol. This study shows that some children can be moved from transfusion to medication after at least a year. The combined understanding and evidence from these two studies brings us closer to achieving the National Institutes' goal of a 'stroke free generation' in sickle cell disease."
Standard treatment for children with sickle cell disease who are at high risk of stroke consists of regular blood transfusions. Children who receive regular blood transfusions are then at risk for iron overload. Chelation, or iron-reduction, therapy is needed for those receiving transfusions. The National Institutes of Health (NIH)-supported study sought to answer whether hydroxyurea would provide the same benefit as blood transfusions, given these additional treatment impacts. Hydroxyurea is the only drug approved by the Food and Drug Administration to treat sickle cell disease. The Transcranial Doppler with Transfusions Changing to Hydroxyurea (TWiTCH) study was stopped early due to positive preliminary results in November 2014.
Researchers from 26 clinical sites supported by the NIH's National Heart, Lung, and Blood Institute (NHLBI) recruited and studied 121 children ages 4 to 16 years old and divided them into two groups: one that received transfusions and one that was transitioned from transfusions to daily doses of hydroxyurea.
"No child should ever have to face the prospect of suffering through a stroke," said Gary H. Gibbons, M.D., director of the NHLBI. "Our institute is striving to achieve a stroke-free generation of children living with sickle cell disease. Studies like this are vital for moving us toward this worthwhile goal."
Study authors indicated that the findings suggest that hydroxyurea could be effective at reducing risk of stroke for other patient populations, though this was not a primary goal of the study.
More information: Russell E Ware et al. Hydroxycarbamide versus chronic transfusion for maintenance of transcranial doppler flow velocities in children with sickle cell anaemia—TCD With Transfusions Changing to Hydroxyurea (TWiTCH): a multicentre, open-label, phase 3, non-inferiority trial, The Lancet (2015). DOI: 10.1016/S0140-6736(15)01041-7