Study finds effect of PrEP on bone density is reversible
The slight loss in bone mineral density associated with HIV pre-exposure prophylaxis (PrEP) antiretroviral use is reversible in young adult patients who stop taking the drugs, according to findings presented by researchers today at the 23rd Conference on Retroviruses and Opportunistic Infections (CROI) in Boston. PrEP is an HIV prevention strategy in which at-risk HIV-negative people take a daily pill of Truvada, which contains the antiretroviral drugs tenofovir and emtricitabine, to prevent them from becoming infected.
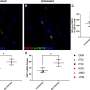
The findings result from a bone mineral density substudy of two large clinical trials, iPrEx and iPrEx OLE, funded by the National Institute of Allergy and Infectious Diseases (NIAID). Data from the substudy presented today illustrate that bone mineral density decreased a measurable but clinically insignificant amount over the course of a year in young adult males and transgender participants with an average age of 24 taking a protective amount of PrEP. However, six months after stopping the regimen, bone mineral density levels in the spines of these individuals increased to levels consistent with study participants of the same age who took a placebo. Hip bone mineral densities also increased in the first six months after stopping PrEP and returned to normal levels by a median follow-up time of 73 weeks.
Previous studies using sensitive scans have shown that HIV medications containing tenofovir slightly reduce bone mineral density, though not to a degree at which patients experience complications. This is the first study to show that this effect is reversible when a patient can stop PrEP, such as when an individual enters into a mutually monogamous relationship with another HIV-negative individual.
Overall, the new findings indicate that Truvada-based oral PrEP does not pose an irreversible effect on bone mineral density and support using PrEP to prevent HIV infection in at-risk young adults.
More information: These findings were presented today at the 23rd Conference on Retroviruses and Opportunistic Infections at the John B. Hynes Veterans Memorial Convention Center in Boston.