Investigating potential fetal exposure to antidepressants
Depression is a serious issue for expecting mothers. Left untreated, depression could have implications for a fetus's health. But treating the disease during pregnancy may carry health risks for the developing fetus, which makes an expecting mother's decision whether to take medication a very difficult one. To better understand how antidepressants affect fetuses during pregnancy, scientists studied exposure in mice. They report their findings in the journal ACS Chemical Neuroscience.
In recent years, the number of pregnant women taking antidepressants has been on the rise. Studies have suggested that common antidepressants known as selective-serotonin reuptake inhibitors (SSRIs) could affect the health of these women's children. How they might do this is unclear. For example, pregnancy could affect the metabolism of SSRIs in the body and in turn a fetus's exposure to them. Alexandre Bonnin and colleagues at the Zilkha Neurogenetic Institute (University of Southern California) wanted to gain more insight into how SSRIs are metabolized and travel in mothers and their fetuses.
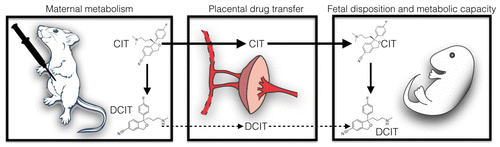
The researchers administered a widely prescribed SSRI, citalopram, to pregnant mice and tracked what happened to the drug. They found that the medication and its primary metabolite travel quickly to fetuses and their brains.
Also, two hours after administrating the drug, the fetuses had higher concentrations of the drug in their blood than their mothers. In addition, they found important differences in fetal drug exposure depending on the stage of pregnancy. Further work will be needed to see whether SSRIs might act similarly in humans and what effect this exposure might have on fetuses.
More information: Juan C. Velasquez et al. Maternal Pharmacokinetics and Fetal Disposition of (±)-Citalopram during Mouse Pregnancy, ACS Chemical Neuroscience (2016). DOI: 10.1021/acschemneuro.5b00287
Abstract
While selective-serotonin reuptake inhibitor (SSRI) antidepressants are commonly prescribed in the treatment of depression, their use during pregnancy leads to fetal drug exposures. According to recent reports, such exposures could affect fetal development and long-term offspring health. A central question is how pregnancy-induced physical and physiological changes in mothers, fetuses, and the placenta influence fetal SSRI exposures during gestation. In this study, we examined the effects of gestational stage on the maternal pharmacokinetics and fetal disposition of the SSRI (±)-citalopram (CIT) in a mouse model. We determined the maternal and fetal CIT serum concentration–time profiles following acute maternal administration on gestational days (GD)14 and GD18, as well as the fetal brain drug disposition. The results show that pregnancy affects the pharmacokinetics of CIT and that maternal drug clearance increases as gestation progresses. The data further show that CIT and its primary metabolite desmethylcitalopram (DCIT) readily cross the placenta into the fetal compartment, and fetal exposure to CIT exceeds that of the mother during gestation 2 h after maternal administration. Enzymatic activity assays revealed that fetal drug metabolic capacity develops in late gestation, resulting in elevated circulating and brain concentrations of DCIT at embryonic day (E)18. Fetal exposure to the SSRI CIT in murine pregnancy is therefore influenced by both maternal gestational stage and embryonic development, suggesting potential time-dependent effects on fetal brain development.













