February 15, 2016 report
Researchers report possible therapeutic approach to fatty liver disease
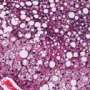
(Medical Xpress)—Steatosis is a condition in which the body fails to properly synthesize or eliminate triglyceride fat, causing abnormal retention of lipids inside cells. Steatohepatitis, then, is liver disease characterized by inflammation and fat accumulation. The condition is typically seen in alcoholics, but it is often observed in people with diabetes and obesity. Ultimately, steatohepatits can progress to life-threatening cirrhosis.
Given the exponential rise of diabetes in developed countries, steatosis and steatohepatitis are subjects of tremendous interest to medical researchers. A new study, published in the Proceedings of the National Academy of Sciences, reports that treatment with fibroblast growth factor 1 (FGF1) is an important factor in adipose remodeling processes—mice deficient in FGF1 displayed a severe diabetic phenotype and increased inflammation and fibrosis in fatty tissues.
FGF1 is a protein encoded by the FGF1 gene. It encourages cell division and expresses a number of potent cell survival traits; this family of proteins is involved in a great number of functions including cell growth, embryonic development and tissue repair. In the current study, the researchers studied two different lines of mice: leptin-deficient mice, which develop steatosis through excessive eating; and mice with a dietary choline deficiency, which develop steatosis as a result of a defect in the way fat is broken down in the liver.
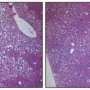
They found that FGF1 had dramatic antisteatotic effects in the leptin-deficient mice. Additionally, in these mice, FGF1 suppressed liver inflammation—hepatic steatosis can often develop into dangerous steatohepatitis characterized by serious inflammation. However, the antisteatotic effects observed in these mice were completely absent in the choline-deficient mice.
Choline deficiency causes steatosis in the absence of metabolic disease such as diabetes or obesity; the researchers conclude that the antisteatotic effects of FGF1 are dependent on catabolic pathways that are defective in the choline-deficient mice. The differences between the lines were also indicated by the zonal distribution of lipids in the animals' livers; lipid zoning is an important activity for segregating different metabolic pathways in the liver.
The study also concludes that FGF1 suppresses liver inflammation completely independently of its effects on fatty liver disease. "Our results show that rFGF1 effectively suppresses hepatic inflammation both in [leptin-deficient] mice and choline-deficient mice, as indicated by significant reductions in the expression of… proinflammatory M1 markers," the authors write.
And although the researchers thought there was potential for the treatment to produce such adverse effects as fibrosis and proliferation, these were not observed.
These results are important because no pharmacological treatments have been approved for steatohepatitis, and there are drawbacks to the existing approaches of insulin sensitizers and antioxidative treatments. By contrast, the study findings conclude that FGF1 improved hyperglycemia, insulin sensitivity and steatosis symptoms in the obese mice, though not the choline deficient line. "Our findings indicate that, in addition to its potent glucose-lowering and insulin-sensitizing effects, rFGF1 could be therapeutically effective in the treatment of non-alcoholic fatty liver disease," the authors write.
More information: Effective treatment of steatosis and steatohepatitis by fibroblast growth factor 1 in mouse models of nonalcoholic fatty liver disease. PNAS 2016 ; published ahead of print February 8, 2016, DOI: 10.1073/pnas.1525093113
Abstract
Nonalcoholic fatty liver disease (NAFLD) is the most common chronic liver disorder and is strongly associated with obesity and type 2 diabetes. Currently, there is no approved pharmacological treatment for this disease, but improvement of insulin resistance using peroxisome proliferator-activated receptor-γ (PPARγ) agonists, such as thiazolidinediones (TZDs), has been shown to reduce steatosis and steatohepatitis effectively and to improve liver function in patients with obesity-related NAFLD. However, this approach is limited by adverse effects of TZDs. Recently, we have identified fibroblast growth factor 1 (FGF1) as a target of nuclear receptor PPARγ in visceral adipose tissue and as a critical factor in adipose remodeling. Because FGF1 is situated downstream of PPARγ, it is likely that therapeutic targeting of the FGF1 pathway will eliminate some of the serious adverse effects associated with TZDs. Here we show that pharmacological administration of recombinant FGF1 (rFGF1) effectively improves hepatic inflammation and damage in leptin-deficient ob/ob mice and in choline-deficient mice, two etiologically different models of NAFLD. Hepatic steatosis was effectively reduced only in ob/ob mice, suggesting that rFGF1 stimulates hepatic lipid catabolism. Potentially adverse effects such as fibrosis or proliferation were not observed in these models. Because the anti-inflammatory effects were observed in both the presence and absence of the antisteatotic effects, our findings further suggest that the anti-inflammatory property of rFGF1 is independent of its effect on lipid catabolism. Our current findings indicate that, in addition to its potent glucose-lowering and insulin-sensitizing effects, rFGF1 could be therapeutically effective in the treatment of NAFLD.
© 2016 Medical Xpress