Which neuron is more mature? Single cell transcriptome knows
The human brain is extremely complex, containing billions of neurons forming trillions of synapses where thoughts, behavior and emotion arise. However, when an individual is performing a particular task, not many but only a few neural circuits are in action. The enormous cellular heterogeneity of the brain structure has made dissections of the molecular basis for neural circuitry function particularly challenging, because previous studies on genetic and epigenetic profiling using a block of brain tissues simply do not have the sufficient precision and accuracy to correspond to the activities of a few activated circuitries in the brain.
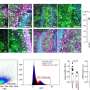
In the March issue of Springer's journal Protein & Cell, Chen et al. reported, for the first time, molecular gene expression signatures underlying human neuronal maturation judged by electrophysiological characteristics. The authors successfully employed "Patch-seq" technology whereby patch-clamp recording and single neuron transcriptome profiling were performed on the same human neurons derived from more primitive stem cells. The authors also implemented powerful bioinformatics analyses, including Weighted Gene Coexpression Network Analyses (WGCNA), to extrapolate important signature genes corresponding to immature and mature human neurons, which is unprecedented.
By cross-referencing published data to single neuron transcriptome profiled from fetal and adult human brains (though without electrophysiological analyses), 39 neuronal genes were identified, which could serve as generic biomarkers for human neuronal maturation. The precise role of these 39 genes in the neuronal maturation process remains to be determined in the future. On the other hand, a number of calcium signaling, mitochondrial function, and ubiquitination-related genes are consistently elevated when human neurons mature, underscoring the critical importance of these biological processes relating to neuronal function. It is expected that dysregulation of these processes might be core elements leading to neurodegeneration.
The technology of coupling electrophysiological recording and single cell transcriptome analysis plus the powerful WGCNA should, in the future, be widely used to delineate the molecular logic for neural circuitry wiring, function and plasticity, to ultimately understand the human brain, the mind, and major human neurological disorders.
More information: Xiaoying Chen et al. Coupled electrophysiological recording and single cell transcriptome analyses revealed molecular mechanisms underlying neuronal maturation, Protein & Cell (2016). DOI: 10.1007/s13238-016-0247-8