Tocilizumab treats rheumatoid arthritis after non-TNFi failure
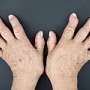
(HealthDay)—For patients with rheumatoid arthritis (RA) and a first non-tumor necrosis factor inhibitor (TNFi) failure, treatment with tocilizumab seems more efficacious than abatacept or rituximab, according to a study published online March 27 in the International Journal of Rheumatic Diseases.
Tristan Pascart, M.D., from Lille University in Lomme, France, and colleagues conducted a retrospective, multicenter study involving patients treated for RA with abatacept, rituximab, or tocilizumab after a non-TNFi failure. Data were collected for 100 patients who started a second non-TNFi between 2006 and 2013 (15 treated with rituximab, 36 with tocilizumab, and 49 with abatacept).
The researchers found that the change in Disease Activity Index of 28 joints-erythrocyte sedimentation rate (DAS28-ESR) differed significantly between the groups (P = 0.001). The decrease in DAS28-ESR was higher for patients treated with tocilizumab versus abatacept (median, 36 versus 0 percent; P = 0.002). A similar difference was seen for tocilizumab and rituximab, although the difference was not significant (median decrease, 36 versus 0 percent; P = 0.07). The results were similar for 12-month change in DAS28-C-reactive protein.
"This study suggests a better efficacy of tocilizumab compared with abatacept and rituximab in situations of non-TNFi failure," the authors write.
Several authors disclosed financial ties to the pharmaceutical and medical technology industries.
More information:
Abstract
Full Text (subscription or payment may be required)
Copyright © 2016 HealthDay. All rights reserved.