Researchers identify a new signaling mechanism implicated in congenital aortic valve disease
Researchers at the Centro Nacional de Investigaciones Cardiovasculares Carlos III (CNIC) have demonstrated the crucial role of the NOTCH signaling pathway in the development of a fundamental heart structure, the heart valves. These valves ensure unidirectional bloodflow with each heartbeat, and open and close millions of times during a person's lifetime. The results, published in Circulation Research, could help to improve the genetic diagnosis and treatment of patients with a bicuspid aortic valve (BAV), a highly prevalent condition (affecting 1-2% of the population) that seriously compromises cardiovascular health.
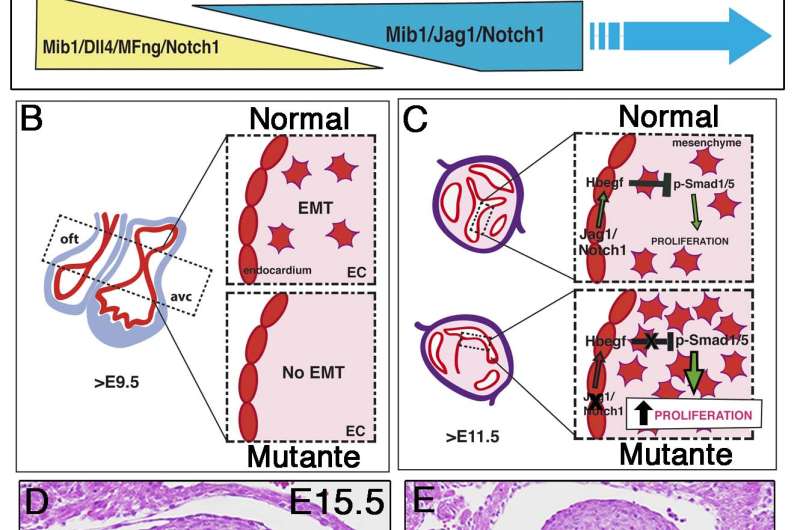
Using genetically modified mouse strains, the research team showed that the NOTCH signaling pathway plays an essential role throughout the process of heart valve formation. NOTCH is required for the formation of the primitive valves that function during fetal development and later for the acquisition of the definitive form of the mature valves. The study identifies a new NOTCH-regulated molecular mechanism that controls proliferation during late valve development. The research team, led by José Luis de la Pompa, showed that this control is inactivated by alterations to genes encoding NOTCH pathway components. The resulting excessive growth results in the fusion of the original 3 valves into 2, producing a bicuspid aortic valve instead of the normal tricuspid valve.
Congenital defect
BAV is the most frequent congenital heart defect and affects 3 times as many men as women. The bicuspid valve is predisposed to premature calcification, and as a result the valve normally needs to be surgically replaced at a relatively young age (30-40 years). First author Donal MacGrogan added that the effects of BAV are not limited to valve formation, but also affect the development of the aorta.
The research findings could have important clinical implications in relation to both early genetic diagnosis and the classification of patients with heart valve malformations, and could contribute to the eventual development of therapies aimed at controlling excessive valve growth.
More information: Sequential Ligand-Dependent Notch Signaling Activation Regulates Valve Primordium Formation and Morphogenesis , circres.ahajournals.org/conten … .115.308077.abstract , DOI: 10.1161/CIRCRESAHA.115.308077