Fighting addiction through the gut-brain axis
"We are what we eat." This piece of folk knowledge is true in more ways than one. In fact, it is well known that food, through action on the enteric system, has direct effects on the brain. The mechanisms underlying this exchange are no longer a mystery since a large body of scientific literature highlights the functional crosstalk between the periphery and the brain. In particular, the so-called gut-brain axis consists of bidirectional communication between the brain and the enteric system, linking the emotional and cognitive centers of the brain with peripheral intestinal functions (and vice versa). Insights into the gut-brain crosstalk have revealed an extremely fascinating communication system that is thought to be involved in many routine functions and systems within the healthy body, as well as in many diseases.
Could this axis be a surprising key player in drug addiction? Recently, a pivotal role of appetite regulatory peptides, such as ghrelin and glucagon-like peptide 1 (GLP-1), in the development of drug reinforcement has been identified (Engel JA and Jerlhag E, 2014). Development of drug dependence is attributed to the ability of addictive drugs to interfere with the mesoaccumbal dopamine system, which consists of dopaminergic neurons of the ventral tegmental area (VTA) projecting to the nucleus accumbens (NAc). Moreover, in a recent PLOS ONE article, Vallof and colleagues observed that Neuromedin U (NMU), another gut-brain peptide, was able to attenuate the psychomotor effects induced by the psychostimulant amphetamine. This finding is of great interest, especially being that pharmacological interventions to treat amphetamine dependence are limited.
How does NMU act on the brain?
Neuropeptide NMU, which is detected throughout the enteric system, acts via two distinct receptors, namely NMUR1 and NMUR2. While NMUR1 is expressed mainly in the peripheral system, NMUR2 is found also in the brain, especially, in reward-related areas. Congruent with the high expression of NMUR2 along the gut-brain axis, research has shown that NMU serves as a catabolic (anorexigenic) signal via a central mechanism (Ida T et al., 2005). In fact, central administration of NMU decreases food intake in rats, whereas NMU antisense increases it. These anorexigenic properties of NMU are further confirmed by the findings that NMU overexpressing mice are hypophagic (Kowalski TJ et al., 2005).
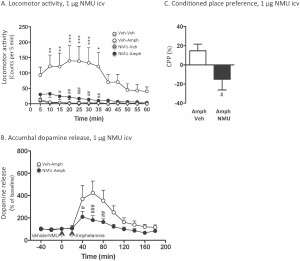
Regarding the NMU-mediated attenuation of amphetamine related effects on the brain, Vallof and colleagues, from the University of Gothenburg in Sweden, showed that central activation of the NMU receptor signaling system inhibited the psychomotor effects induced by amphetamine administration. In particular, activation of the central NMU receptor was able to reduce dopamine release in the nucleus accumbens and amphetamine-induced hyperlocomotion, thus suggesting that NMU may act by interfering with dopamine release. This hypothesis was further supported by microdialysis experiments showing a reduction in dopamine release upon NMU administration.
In fact, when rats were intravenously injected with NMU before amphetamine administration, they were less responsive to the stimulant effects of the recreational drug. Finally, in order to investigate the therapeutic effect of NMU on amphetamine dependence, the authors performed a conditioned-place preference test (CPP), a paradigm designed to reveal the pro- or the anti-addictive properties of a given compound. CPP constitutes a measure of reward-related contextual cues associated with a given drug experience. Interestingly, rats pre-treated with NMU did not show expression of conditioned place preference, thus revealing that NMU may counterbalance the addictive effects of amphetamine.
Central administration of NMU attenuates amphetamine-induced locomotor stimulation, accumbal dopamine release and expression of conditioned place preference in mice.
The above findings lead to a critical question: which brain region is involved in NMU action? The nucleus accumbens, a key region of the reward system, was the obvious candidate. Surprisingly, even though NMU injected in the nucleus accumbens was able to strongly reduce amphetamine-evoked psychomotor effects, it failed to prevent conditioned place preference. These results suggest a complex cerebral mechanism by which the gut peptide may act on the reward system. Elisabet Jerhalg, the principal investigator of the study, highlighted the importance of considering "that NMUR2 receptors expressed in other brain areas, such as the arcuate nucleus and paraventricular nucleus, are important for amphetamine-induced activation of the mesoaccumbal dopamine system". The authors went on to say that "the downstream mechanisms through which central NMU signaling reduces amphetamine-induced reward remain unknown and need to be further investigated". An interesting hypothesis on the mechanism of action of NMU resides on the stress axis. Indeed, Vallof and colleagues speculated on "the possibility that NMU [mediated attenuation of] amphetamine-induced reward [could be] secondary to its effect on the hypothalamus-pituitary-adrenal (HPA) stress axis". Paradoxically, this hypothesis would entail that "NMU mediated reward does not involve stress response since it has been shown that the selected dose, as opposed to higher doses, of … intravenous NMU does not affect corticosterone levels [which are usually elevated in stress-like responses] in rodents," continued the authors.
In spite of these promising results, the functions of the neuropeptide NMU in the brain still remain a mystery, which deserve to be fully explored. Who knows, the gut-brain axis might contain several surprises that need to be unraveled.
More information: Jörgen A. Engel et al. Role of Appetite-Regulating Peptides in the Pathophysiology of Addiction: Implications for Pharmacotherapy, CNS Drugs (2014). DOI: 10.1007/s40263-014-0178-y
Ida T, Mori K, Miyazato M, Egi Y,Abe S, Nakahara K, et al. Neuromedin is a novel anorexigenic hormone. Endocrinology. 2005;146(10):4217–23. PMID: 15976061
Kowalski TJ, Spar BD, Markowitz L, Maguire M, Golovko A, Yang S, et al. Transgenic overexpression of neuromedin U promotes leanness and hypophagia in mice. The Journal of endocrinology. 2005; 185 (1):151–64. PMID: 15817836
This story is republished courtesy of PLOS Blogs: blogs.plos.org.