Diagnosing Alzheimer's earlier rather than later
A hallmark of Alzheimer's disease is the appearance of plaques in the brain. The plaques are gradually made up by the aggregation of a small protein called amyloid-beta or "Abeta". Alzheimer's is usually diagnosed late, when the plaques have developed and are already causing cognitive damage like memory loss. In a large study led by EPFL, an international team of scientists from Harvard, Edinburgh and Ulm, have now found a way to detect Abeta aggregation in its early stages, which will make diagnosing and treating the disease much more effective. The study is published in the journal Alzheimer's and Dementia.
Alzheimer's – and a number of other diseases – begins when Abeta proteins begin to aggregate, or clump together, in the brain and form larger structures called oligomers ("oligo" = few). The oligomers themselves then clump together to form the even larger amyloid plaques. The problem is that the molecular mechanisms that lead single Abeta to clump into oligomers early on are not well studied, and even the exact structure of the oligomers is a matter of debate among scientists.
Scientists at EPFL's Protein Core Facility, have now led an international team of scientists from Harvard, Edinburgh and Ulm in one of the most extensive analysis of Abeta peptides. The project aimed to understand what happens during aggregation, and particularly at its early stages.
Using different analysis techniques (e.g. mass spectrometry), the researchers identified specific points on Abeta where it undergoes a process called "autocleavage". Normally, Abeta is cut by specialized enzymes as part of the cell's cleaning process. But, in this case, Abeta simply breaks at very specific points along its structure without any enzymes being involved.
The researchers found that Abeta autocleavage happens during the early stages of aggregation. The process generates truncated Abeta fragments, which can then form complexes with normal, unbroken Abeta molecules. This produces the neurotoxic oligomers that are thought to cause the early cognitive problems of Alzheimer's.
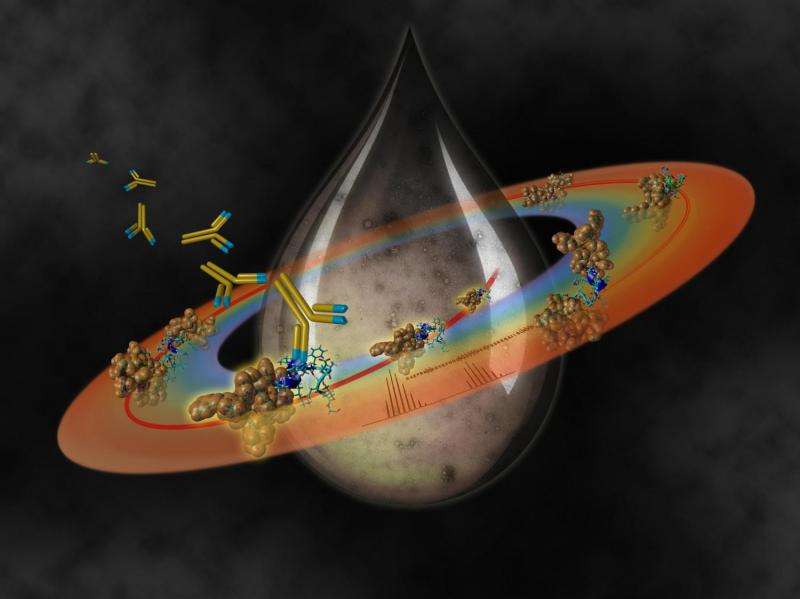
In addition, the scientists found that autocleavage of Abeta also produces a pattern or "fragment signature" that is highly reproducible, meaning that it can be used to detect Abeta aggregation early on.
The research team also screened mice that are used as standard models of Alzheimer's disease, as well as human post-mortem brain tissue and cerebrospinal fluid. The investigation showed that the process of autocleavage, (as a result of Abeta aggregation), is a highly relevant clinical feature of Alzheimer's.
Finally, the researchers also found that the truncated peptides can be used to develop antibodies that can identify and bind them. This would enable doctors to monitor the accumulation of truncated Abeta fragments in diagnostic tools, and even target them for clearing by the patient's immune system.
"The study reveals novel Abeta targets associated with the early events of oligomerization," says Adrian Schmid at EPFL's Protein Core Facility and senior author of the paper. "This may help define the preclinical stage of Alzheimer's, and determine the factors that predict the emergence of clinical impairment and progression to severe dementia."
The findings take our knowledge about Abeta aggregation to a new level and have important implications in Alzheimer's diagnosis and future therapies. "Preclinical biomarkers of dementia can help identify individuals who may benefit from early therapeutic intervention," says Schmid. "It can therefore improve the patient's overall quality of life while reducing the costs associated with long-term medical care."
More information: Nikita Rudinskiy et al. Amyloid-beta oligomerization is associated with the generation of a typical peptide fragment fingerprint, Alzheimer's & Dementia (2016). DOI: 10.1016/j.jalz.2016.03.011