Early warning: Current Japanese encephalitis vaccine might not protect
Japanese encephalitis virus (JEV) is the leading cause of viral encephalitis (infection of the brain) in Asia. There is no specific treatment for Japanese encephalitis (JE) which can cause death or serious long-term disability, and WHO recommends JEV vaccination in all areas where the disease is recognized as a public health priority. A study published in PLOS Neglected Tropical Diseases suggests that current vaccines may fail to protect individuals against an emerging strain of the virus.
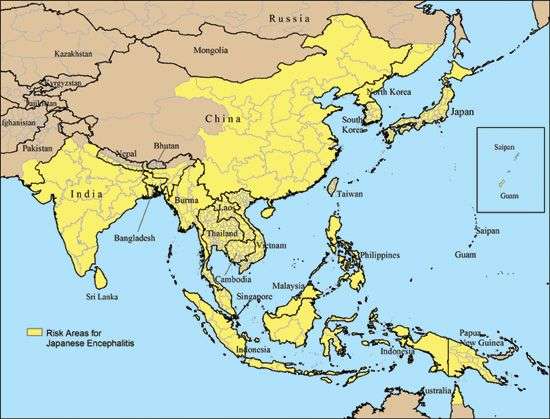
An estimated 3 billion people live in 24 South-East Asian and Western Pacific countries where the virus is present. JE viruses come in different 'flavors': there are five different genotypes (G1-G5), defined by differences in the 'envelope' gene that codes for proteins covering the virus surface. Strain G5 was originally isolated from a patient and described in 1951, but then not seen again until found recently (in 2009) in China and subsequently in Korea.
No specific treatment exists against the JE virus, but a number of vaccines are used to protect local populations and travellers. All of the vaccines are based on G3 virus strains and have been shown to work well against G1 through G4 strains. However, their efficiency against the previously rare but possibly re-emerging G5 strain is not clear.
Guodong Liang, from the Chinese Center for Disease Control and Prevention, in Beijing, China, and colleagues were the first to report the re-emergence of the G5 strain. In this study, they compared G3 and G5 viruses and tested whether the vaccine commonly used in China can protect against G5 viruses.
Having found the two strains similar in their ability to cause disease in mice, the researchers vaccinated mice and tested whether they were protected against a dose of virus that would be lethal to unvaccinated animals. They found that the (G3-based) vaccine protected all the mice against a lethal challenge with G3 virus, but only 50% of the mice infected with G5 virus survived.
Next, the researchers looked for inactivating (or neutralizing) antibodies in vaccinated two-year-old children. They examined blood samples from 26 children that had been collected both before and 28 days after JE vaccination. Following vaccination, they were able to detect neutralizing antibodies against G3 strains in all the children, but only 35% of them also had antibodies that could neutralize G5 strains.
Finally, the researchers asked whether people who had been infected with JEV naturally (presumably with strains other than G5) and developed encephalitis had antibodies that could neutralize either G3 or G5 strains. Analyzing samples from 45 clinically diagnosed JE patients, they found that while all of the patients had neutralizing antibodies against G3 strains, only 29 of the 45 patients (64%) had the ability to neutralize G5 strains. Most of the latter were older patients; less than half of the pediatric patients (those under age 15) had neutralizing antibodies against G5 virus.
These results suggest that the existing vaccines provide only partial protection against G5 JEV strains. Moreover, natural infection with a different strain might not protect against subsequent G5 infection, especially in children.
As the researchers discuss, whether the JE cases that occurred over recent years despite wide-spread vaccination programs in countries like China and Korea are caused by G5 strains is not known. Nor is it clear how much of a public health threat G5 strains are at present, or might become in the future. Nonetheless, the results reported represent early warning signs of a potential infectious disease crisis in South-East Asia, and further research on the G5 JEV strains and on vaccines that better protect against them seems warranted.
More information: PLOS Neglected Tropical Diseases, DOI: 10.1371/journal.pntd.0004686