Vaccination under the influence of estradiol increases vaginal antiviral immunity
When it comes to sexually transmitted infections (STIs), some female hormones are protective while others make women more susceptible. A study in mice published on May 5th in PLOS Pathogens suggests that estradiol (E2) exerts its protective effect against herpes virus by shifting the immune response in the vaginal mucosa toward a more effective antiviral one.
Many studies have shown that injectable contraceptives containing progestins may increase a woman's risk of being infected with HIV and with HSV-2, the virus causing genital herpes. On the other hand, estradiol, another hormone that is present during the normal menstrual cycle and contained in oral contraceptives, has been shown to be protective. To minimize unintended negative consequences of hormonal contraception, understanding how different hormones affect susceptibility to STI pathogens is important.
Charu Kaushic and colleagues from McMaster University in Hamilton, Canada, had previously shown that vaccination of mice against genital herpes (HSV-2) under the influence of estradiol leads to enhanced protection against the virus. In this study, the researchers examined the underlying mechanisms.
As in the earlier work, they implanted either E2-releasing pellets or control pellets into female mice whose ovaries (which normally produce both estradiol and progesterone) had been removed. The pellets release E2 for three weeks. Following implantation, the researchers exposed the mice to two rounds (2 weeks apart) of nasal HSV-2 vaccine, followed by a vaginal challenge with a high dose of intact HSV-2 virus 6 weeks later.
Most of the mice with the E2 pellets survived the challenge and showed much less severe disease symptoms. In contrast, most of the control mice got seriously sick and were euthanized. This was true for several different HSV vaccine formulations tested.
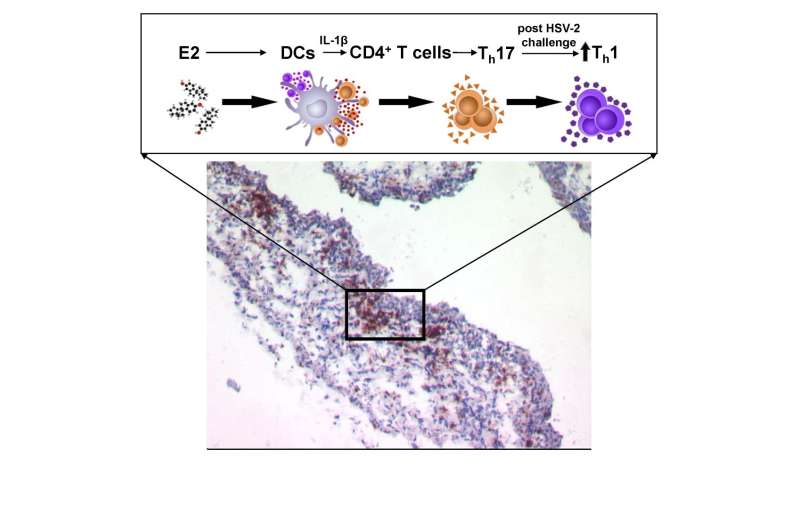
To examine the mechanism by which E2 enhanced immune protection in the HSV-2 vaccine model, the researchers studied immune cells and immune signaling molecules in the female genital tract following the HSV-2 challenge. They found that E2 treatment accelerates and increases the response of two types of T helper cells, namely Th1 and Th17 cells. (Th1 cells are required for host defense against intracellular viral and bacterial pathogens. Th17 cells are defined by their production of IL-17 and promote inflammation.)
Following viral challenge, cells of both types are recruited earlier and at larger numbers to the vaginal mucosa in mice that were vaccinated under E2 influence. Searching for the local immune cells responsible, the reserchers found that specific antigen-presenting cells called dendritic cells were the ones that primarily induced Th17 responses: depletion of vaginal dendritic cells, they saw, decreased the ability of vaginal tissue to promote production of IL-17 in T helper cells, suggesting reduced differentiation into Th17 cells.
This ability of vaginal dendritic cells to induce predominantly Th17 responses was not seen in antigen-presenting cells from spleen, lung, and intestine, suggesting that the hormonal conditioning is unique to the genital mucosa.
Given the increased Th17 responses associated with E2 treatment seen in various experiments, the researchers tested whether IL-17—the immune mediator produced by Th17 cells—played a role in anti-viral immunity against HSV-2. Using "knock-out" mice that lacked IL-17, they found that indeed, IL-17 appears to enhance the anti-viral response by Th1 cells in the vaginal mucosa.
Their study, the researchers summarize, "describes a mechanism by which E2 enhances anti-viral protection following vaccination in the genital HSV-2 mouse model". They add, "to the best of our knowledge, this is the first study demonstrating that E2 can directly regulate T-cell mediated adaptive anti-viral immunity in the female genital tract by modulating DC functions".
More information: Anipindi VC, Bagri P, Roth K, Dizzell SE, Nguyen PV, Shaler CR, et al. (2016) Estradiol Enhances CD4+ T-Cell Anti-Viral Immunity by Priming Vaginal DCs to Induce Th17 Responses via an IL-1-Dependent Pathway. PLoS Pathog 12(5): e1005589. DOI: 10.1371/journal.ppat.1005589