Canada to hold first Zika vaccine test on humans
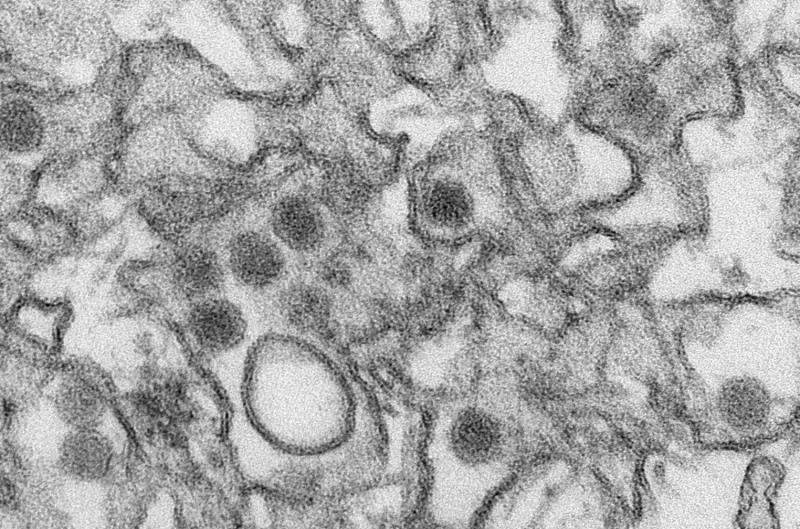
A Canadian university said Tuesday it will conduct the world's first Zika vaccine test on humans as health authorities scramble to counter the explosively spreading virus.
The Zika vaccine that's under development will be administered to humans "in the coming days," Universite Laval, based in Quebec City, said in a statement.
"We're very proud to be part of the first international team in the world to complete all of the steps in the regulatory process," said Gary Kobinger, doctor of microbiology and professor of medicine at the university, who is overseeing the study.
Kobinger noted that development of the Zika vaccine was authorized by the US Food and Drug Administration and Health Canada. Until now, vaccines have been tested on mice.
Zika virus is transmitted primarily by mosquitoes and there is no existing treatment or vaccine for it. The virus is capable of causing serious birth defects and has traveled quickly through Latin America.
Kobinger, a global authority on vaccines, is a researcher at CHU, a hospital center affiliated with the university. CHU is conducting the study in collaboration with two centers in the United States.
"CHU de Quebec-Universite Laval is one of the three leading research centers involved in the vaccine study and we're very proud of that," said Gertrude Bourdon, president and CEO of CHU.
More than a dozen pharmaceutical companies, including France's Sanofi and India's Bharat Biotech, are working on developing a Zika vaccine, according to the World Health Organization.
The WHO has forecast sharp growth in the outbreak in the Americas, with as many as four million people infected.
© 2016 AFP













