Gene therapy in a droplet could treat eye diseases, prevent blindness
Eye diseases such as diabetic retinopathy and age-related macular degeneration are among the leading causes of irreversible vision loss and blindness worldwide. Currently, gene therapy can be administered to treat these conditions—but this requires an injection. Now researchers report in the journal ACS Applied Materials & Interfaces a new way to deliver the treatment topically, without a needle.
The rear part of the eye is where conditions such as proliferative diabetic retinopathy and age-related macular degeneration develop. And they involve a substance called vascular endothelial growth factor, which stimulates the growth of blood vessels. Scientists have been trying to inhibit the growth factor using gene therapy, but delivering drugs to the back of the eye currently requires an injection. Not surprisingly, few patients opt for this type of treatment. Gang Wei and colleagues wanted to find a noninvasive approach.
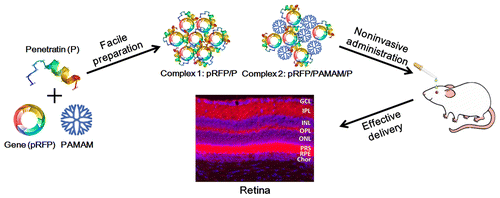
The researchers developed a gene delivery system with a peptide called penetratin, which has shown good permeability in the eye, and a synthetic polymer called poly(amidoamine) that has previously been used in drug delivery. Testing on rats showed that the complex, when applied as an eye drop, rapidly moved from the eye's surface to its rear inner lining. It remained in the retina for more than eight hours—enough time for a model gene to be expressed. The findings demonstrate the method's potential for delivering gene therapy to treat several eye diseases, the researchers say.
More information: Chang Liu et al. Facile Noninvasive Retinal Gene Delivery Enabled by Penetratin, ACS Applied Materials & Interfaces (2016). DOI: 10.1021/acsami.6b04551
Abstract
Gene delivery to the posterior segment of the eye is severely hindered by the impermeability of defensive barriers; therefore, in clinical settings, genomic medicines are mainly administered by intravitreal injection. We previously found that penetratin could transport the covalently conjugated fluorophore to the fundus oculi by topical instillation. In this study, gene delivery systems enabled by penetratin were designed based on electrostatic binding to target the retina via a noninvasive administration route and prepared with red fluorescent protein plasmid (pRFP) and/or poly(amidoamine) dendrimer of low molecular weight (G3 PAMAM). Formulation optimization, structure confirmation, and characterization were subsequently conducted. Penetratin alone showed limited ability to condense the plasmid but had powerful uptake and transfection by corneal and conjunctival cells. G3 PAMAM was nontoxic to the ocular cells, and when introduced into the penetratin-incorporated complex, the plasmid was condensed more compactly. Therefore, further improved cellular uptake and transfection were observed. After being instilled in the conjunctival sac of rats, the intact complexes penetrated rapidly from the ocular surface into the fundus and resided in the retina for more than 8 h, which resulted in efficient expression of RFP in the posterior segment. Intraocular distribution of the complexes suggested that the plasmids were absorbed into the eyes through a noncorneal pathway during which penetratin played a crucial role. This study provides a facile and friendly approach for intraocular gene delivery and is an important step toward the development of noninvasive gene therapy for posterior segment diseases.