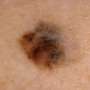
pCR of 37 percent for topical imiquimod in lentigo maligna

(HealthDay)—For patients with lentigo maligna (LM) in-situ melanoma, topical imiquimod has a pathological complete regression (pCR) rate of 37 percent, according to a study published online Oct. 6 in the British Journal of Dermatology.
Jeremy R. Marsden, M.D., from the University Hospital Birmingham NHS Foundation Trust in the United Kingdom, and colleagues estimated pCR for LM following imiquimod in a single-arm phase II trial. Data were included for 60 imiquimod applications over 12 weeks for LM, then radical resection.
The researchers found that the pCR rate was 37 percent (10/27). Twelve of 28 patients had clinical complete remission plus negative biopsies, of whom seven out of 11 had pCR on subsequent surgery. The median dose intensity was 86.7 percent. If the cure rate for imiquimod was 80 percent, eight of 16 surveyed patients preferred primary imiquimod over surgery compared with four of 16 patients if the cure rate was ≤50 percent.
"The pCR rate was insufficient to justify phase III investigation of imiquimod versus surgery. Clinical complete response and negative targeted biopsies left uncertainty regarding pathological clearance," the authors write. "Some patients would trade less aggressive treatment of LM against efficacy."
More information: Full Text (subscription or payment may be required)
Copyright © 2016 HealthDay. All rights reserved.