Smart drug clears fat from liver and blood
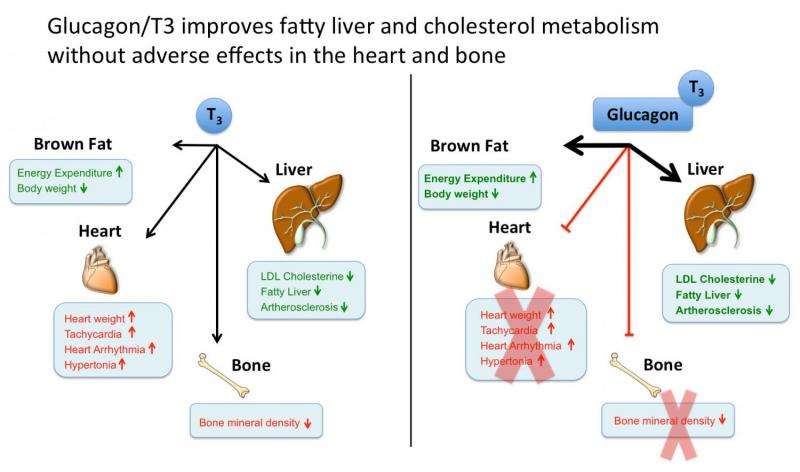
Scientists of Helmholtz Zentrum München and Technische Universität München have developed a 'smart' drug that safely clears the liver of fat and prevents blood vessels from clogging up. Similar to a trojan horse, the drug enters the liver with a trick: It uses the pancreatic hormone glucagon as vehicle to shuttle thyroid hormone T3 the live while keeping it away from other organs, thereby improving cholesterol and lipid metabolism while avoiding typical side effects of thyroid hormone.
The constant rise in obesity and diabetes represents a major burden of our society. Fatty liver and atherosclerosis are frequent consequences of these metabolic diseases, but an efficient and safe medicine, which would reverse obesity, insulin resistance, fatty liver and atherosclerosis remains a major scientific challenge of global priority.
An international team led by metabolism experts Matthias Tschöp (Helmholtz Zentrum München / Technische Universität Müchen), Richard diMarchi (Indiana University) and Timo Müller (Helmholtz Zentrum München) report in the current issue of the journal Cell that liver-specific delivery of the thyroid hormone T3 using glucagon corrects obesity, glucose intolerance, fatty liver disease and atherosclerosis without causing adverse effects in other tissues. "While the ability of T3 to lower cholesterol is known for centuries, deleterious effects, in particular on the skeleton and the cardiovascular system, do so far limit its medicinal utility," says Brian Finan, the first author of the manuscript.
Toward precision medicines of the future
"Part of our trick is, that we use the pancreatic hormone glucagon as a vehicle to deliver thyroid hormone only into cells carrying a glucagon receptor," says Christoffer Clemmensen, who led several of the key experiments. He explains: "Since there are lots of glucagon receptors in the liver, but almost none in heart or bone, our molecule concentrates thyroid hormone action to the liver while keeping it away from places where it would be harmful."
"The next task is to see whether this drug candidate will reach the same level of targeted tissue-selectivity in clinical studies," says diMarchi. "If the molecule shows equal efficacy and safety in humans, then this particular 'smart' drug design may indeed offer perspectives for metabolic precision medicine," summarizes Tschöp.
The team of Matthias Tschöp (Director of the Helmholtz Diabetes Center and Professor for Metabolic Diseases at the Technische Universität München) and Richard DiMarchi (Indiana University) already reported in 2015 the development and evaluation of a single hormone triple agonist, which effectively corrects obesity and insulin resistance. In another study (with a similar working mechanism as in this study) the scientists showed the targeted delivery of estrogen to only cells that express the receptor for GLP-1, thereby improving systems metabolism by concentration the action of estrogen to only the hypothalamus and the pancreas.
The newly developed glucagon/T3 molecule delivered the T3 selectively to the liver and thereby safely improved within a few days cholesterol metabolism in diet-induced obese mice. The molecule further decreased body weight, corrected non-alcoholic fatty liver disease, and improved glucose metabolism without deleterious effects of T3 in the heart and bone. Notably, the molecule failed to improve metabolism in mice lacking either the glucagon receptor or which lack the thyroid hormone receptor in only the liver, demonstrating the liver-specific signal specificity of this new molecule.
More information: Brian Finan et al. Chemical Hybridization of Glucagon and Thyroid Hormone Optimizes Therapeutic Impact for Metabolic Disease, Cell (2016). DOI: 10.1016/j.cell.2016.09.014