Zika virus infection alters human and viral RNA
Researchers at University of California San Diego School of Medicine have discovered that Zika virus infection leads to modifications of both viral and human genetic material. These modifications—chemical tags known as methyl groups—influence viral replication and the human immune response. The study is published October 20 by Cell Host & Microbe.
"I'm excited about this study because it teaches us something new about the human immune system," said senior author Tariq Rana, PhD, professor of pediatrics at UC San Diego School of Medicine. "But these findings are also something researchers should keep in mind as they are designing new Zika virus vaccines and treatments that target the viral genome—some approaches won't work unless they take methylation into account."
In human cells, RNA is the genetic material that carries instructions from the DNA in a cell's nucleus out to the cytoplasm, where molecular machinery uses those instructions to build proteins. Cells can chemically modify RNA to influence protein production. One of these modifications is the addition of methyl groups to adenosine, one of the building blocks that make up RNA. Known as N6-methyladenosine (m6A), this modification is common in humans and other organisms.
In contrast to humans, the entire genomes of some viruses, including Zika and HIV, are made up of RNA instead of DNA. These viruses hijack the host's cellular machinery to translate its RNA to proteins. Rana and his team previously discovered that m6A plays an important role in HIV infection.
"After that, we decided to investigate m6A RNA in Zika virus as well, since we didn't want to miss out on this important information the way we missed it for 30 years of HIV research," Rana said.
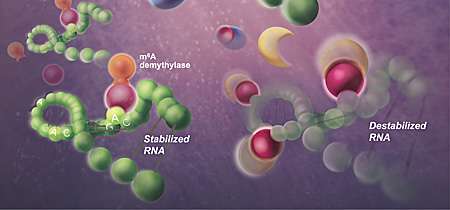
When Zika virus infects a human cell, Rana's team found, the cell modifies viral RNA with m6A as a means to get rid of the infection. RNA tagged with m6A is a beacon for human enzymes that come along and destabilize it. In addition, they found that this host response to Zika viral infection also induced specific m6A modifications on human RNA. These human RNA changes were not present in the absence of Zika virus.
To unravel the role of m6A in Zika virus infection of human cells growing in the laboratory, the researchers removed the human enzymes responsible for adding methyl groups to viral RNA. Without m6A, the viral RNA was more stable and viral replication increased, as compared to human cells with normal methylation enzymes. In contrast, silencing the human enzymes that remove methyl groups—increasing m6A methylation, in other words—decreased Zika virus production.
Next, Rana and team will investigate the role of RNA modifications in the viral life cycle, and how the human immune response is altered by various Zika virus strains. They are also developing small molecules to target specific RNA structures as a means to treat Zika virus infections.
More information: Cell Host & Microbe, DOI: 10.1016/j.chom.2016.10.002