Scientists develop computer models to predict cancer cell network activity
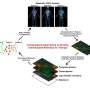
A multi-institution academic-industrial partnership of researchers led by Case Western Reserve University School of Medicine has developed a new method to broadly assess cell communication networks and identify disease-specific network anomalies. The computer-based method, called InFlo, was developed in collaboration with researchers at Philips and Princeton University and predicts how cells send signals across networks to cause cancer or other disease. Details about the new method were recently published in Oncogene.
"Cellular signaling networks are the mechanisms that cells use to transfer, process, and respond to biological information derived from their immediate surroundings," said Vinay Varadan, PhD, assistant professor at Case Western Reserve University School of Medicine, member of the Case Comprehensive Cancer Center, and senior corresponding author on the study. "InFlo can be viewed as modeling the flow of information within these signaling networks."
InFlo works by assessing gene activity levels in tissue samples and predicting corresponding protein levels. It then uses statistical probabilities and other mathematical models to build activity webs showing how the proteins interact. Researchers can use InFlo to compare diseased and healthy tissues and pinpoint signaling differences. InFlo is tissue-specific and accounts for genetic alterations associated with disease, unlike other methods. It represents a major step forward in deciphering the activities of multi-tiered signaling networks commonly used by cells.
"Complex diseases such as cancer involve the simultaneous disruptions of multiple cellular processes acting in tandem," said Varadan. "We developed InFlo to robustly integrate multiple molecular data streams and develop an integrative molecular portrait of an individual cancer sample."
InFlo incorporates data related to each level of cell communication within a single sample, including DNA, RNA, proteins, and molecules commonly attached to proteins such as chemical methyl groups. The method also includes strategies to reduce "noise" and only highlight the signaling networks most likely to cause disease.
Analisa DiFeo, PhD, senior co-corresponding author on the study, Norma C. and Al I. Geller Designated Professor of Ovarian Cancer Research at Case Western Reserve University School of Medicine, and member of the Case Comprehensive Cancer Center, validated InFlo using ovarian cancer tumor cells that were resistant to platinum-based chemotherapy. InFlo pinpointed the interaction between two proteins called cAMP and CREB1 as a key mechanism associated with platinum resistance.
"Following up on InFlo's predictions, we showed that inhibiting CREB1 potently sensitizes ovarian cancer cells to platinum therapy and is also effective in killing ovarian cancer stem cells. We are therefore excited about this discovery and are currently evaluating whether this could lead to a potential therapeutic target for the treatment of platinum-resistant ovarian cancer," said DiFeo.
InFlo is being incorporated into Philips IntelliSpace Genomics platform, and will soon be available for widespread use in basic and translational research settings. Case Western Reserve University researchers will continue to develop the IntelliSpace Genomics InFlo module and the next step will be to expand InFlo to incorporate other data streams. "We are currently collaborating with the Imaging Informatics research group in the Center for Computational Imaging and Personalized Diagnostics at Case Western Reserve University to integrate InFlo with imaging-features derived from pathology and radiology data," said Varadan. Such an addition would result in one of the most comprehensive tools available to researchers to infer mechanisms underlying complex diseases such as cancer.
More information: N Dimitrova et al, InFlo: a novel systems biology framework identifies cAMP-CREB1 axis as a key modulator of platinum resistance in ovarian cancer, Oncogene (2016). DOI: 10.1038/onc.2016.398