Advance in understanding the disparity in prognosis between men and women in melanoma
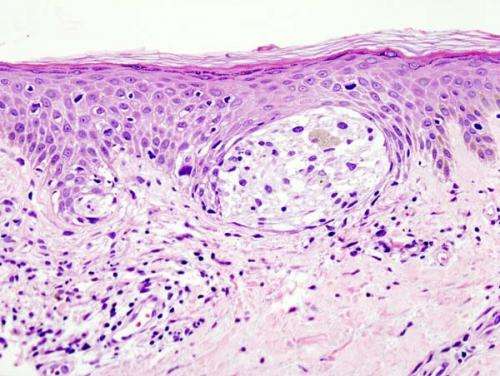
Though it has been shown that men who get melanoma are twice as likely to die from the disease as women, the biological explanation for this is poorly understood. Research led by Dr. Alan Spatz, Director of Surgical and Molecular Pathology at the Jewish General Hospital and head of the "X chromosome and cancer" lab at the Lady Davis Institute in Montreal, reveals that the decreased expression of the X chromosome gene PPP2R3B and its protein PR70 are positively linked to tumor progression in this aggressive form of skin cancer. The finding is published in Science Translational Medicine.
"We focused our research on the machinery of the X chromosome because we postulated that the inactivation of one of the two X chromosomes in women, as opposed to men who have an X and a Y, and the way this mechanism is regulated, may have deep implications on the cell biology of cancer cells" explains Dr. Spatz, who has a long-standing interest in the role of the X chromosome in cancer. "I proposed in 2004 a theoretical model of X-linked tumor suppressor genes loss and oncogenes activation that since has been validated in many situations. The fact that two X's interact together in females has also implications for cancer. We believe that the genetic specificity of the X chromosome plays a significant function in the gender difference we observe in melanoma. And we see unique regulation of tumor suppressor genes and oncogenes in the X chromosome."
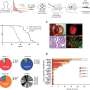
Dr. Spatz, Professor of Pathology and Oncology at McGill University, and Dr. Leon van Kempen, COO and Scientific Director Molecular Pathology Center, have extensively studied the gene PPP2R3B, which is located on the X chromosome in females, but on the Y in males. The expression of this gene has been independently correlated with more favourable progression in melanoma and is important because its expression is higher in females. PPP2R3B codes for the PR70 protein, which decreases melanoma growth by negatively interfering with DNA cell replication and, therefore, acting as an X-linked tumor suppressor.
PR70 is at the forefront of controlling the cell replication cascade. From a clinical perspective, this research suggests there could be potential anti-cancer therapies in actioning the proteins linked to PR70. The proteomics capabilities that have been developed at the Segal Cancer Centre and Lady Davis Institute at the Jewish General Hospital will eventually be instrumental in profiling and identiying the active proteins that could be most effectively targeted by novel therapies.
"I believe this discovery advances our understanding of the specific role of the X chromosome genetics in modulating the expression of genes that are critical in cancer progression," Dr. Spatz said. "Specifically, this is a new avenue for exploring X-linked tumor suppressor genes and oncogenes. I'm confident that we will eventually be able to exploit this discovery to pursue new therapeutic avenues against cancer."
More information: "The protein phosphatase 2A regulatory subunit PR70 is a gonosomal melanoma tumor suppressor gene" by Drs. Leon van Kempen, Alan Spatz et al appears in Science Translational Medicine. stm.sciencemag.org/lookup/doi/ … scitranslmed.aai9188