Antibody combination puts HIV on the ropes
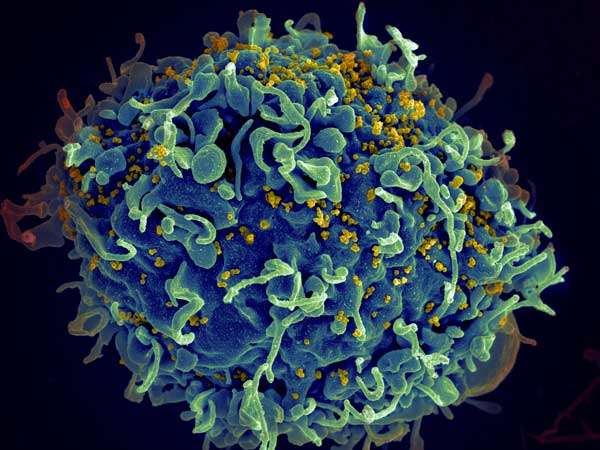
Without antiretroviral drug treatment, the majority of people infected with HIV ultimately develop AIDS, as the virus changes and evolves beyond the body's ability to control it. But a small group of infected individuals—called elite controllers—possess immune systems capable of defeating the virus. They accomplish this by manufacturing broadly neutralizing antibodies, which can take down multiple forms of HIV.
Now a study using antibodies from one of these elite controllers has shown that a combination of three such antibodies can completely suppress the virus in HIV-infected mice. The findings, from the laboratory of Michel Nussenzweig, who is Zanvil A. Cohn and Ralph M. Steinman Professor at Rockefeller University and head of the Laboratory of Molecular Immunology, are being reported in Science Translational Medicine.
"Some people with HIV produce these antibodies, but most of the time the virus eventually escapes them through mutations in the antibody's corresponding epitope," says postdoctoral fellow Natalia Freund, the study's first author. The epitope is the part of the virus that antibodies recognize and attach themselves to, and this ability to mutate makes HIV particularly tricky to tame. It ensures that once the virus is in their bodies, people remain infected forever, and this may be the biggest roadblock in developing immune therapies to overcome the virus.
Tug of war
"Think of the relationship between the antibodies and the virus as an arms race that goes on and on," Freund says. "By mutating, some of the virus may escape the antibodies and continue growing. Years later, the body may produce new broadly neutralizing antibodies against the escaped virus, which in turn may mutate and escape yet again."
"What we've shown in this study is that after several rounds of escape from these particular antibodies, the virus seems to run out of options," she adds. "In this particular case, HIV eventually loses this arms race."
An elite controller's immune system can defeat the virus by coming up with new broadly neutralizing antibodies, and also by producing cytotoxic T cells—immune cells that can recognize and destroy infected cells to immobilize the virus. The patient whose HIV response created antibodies for the study has been working with the Rockefeller team for ten years, contributing his blood serum for their research. He was infected at least three decades ago, and has developed three different types of broadly neutralizing antibodies that bind to three different sites on the virus.
The remarkable thing about his antibodies is that they seem to complement each other's activity, completely shutting down HIV.
The investigators gave the three antibodies, called BG18, NC37, and BG1, to HIV-infected mice whose immune systems had been modified to more closely resemble those of humans. They found that the trio rendered the virus undetectable in two-thirds of the mice three weeks after it was administered.
"This study validates the approach of using three different antibodies to control HIV infection," Freund concludes, "pointing the way toward a potential new treatment for people infected with HIV."
More information: Natalia T. Freund et al, Coexistence of potent HIV-1 broadly neutralizing antibodies and antibody-sensitive viruses in a viremic controller, Science Translational Medicine (2017). DOI: 10.1126/scitranslmed.aal2144