This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Novel vaccine concept generates immune responses that could produce multiple types of HIV neutralizing antibodies
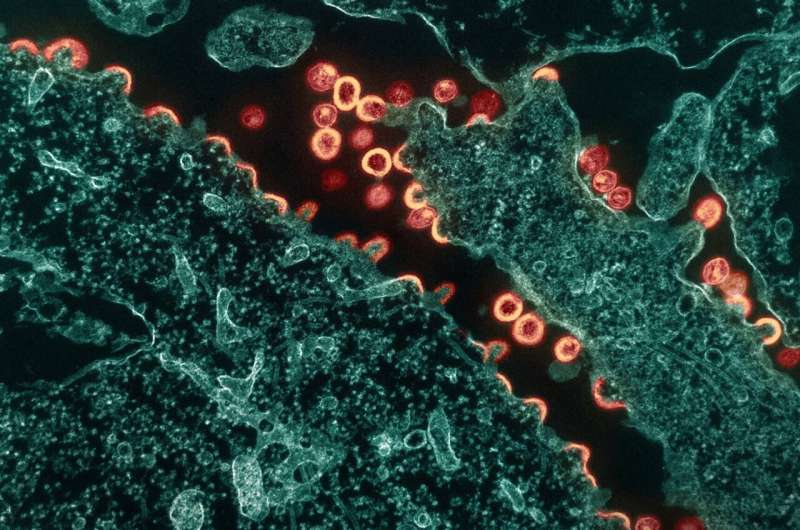
Using a combination of cutting-edge immunologic technologies, researchers have successfully stimulated animals' immune systems to induce rare precursor B cells of a class of HIV broadly neutralizing antibodies (bNAbs). The findings, published today in Nature Immunology, are an encouraging, incremental step in developing a preventive HIV vaccine.
HIV is genetically diverse, making the virus difficult to target with a vaccine, but bNAbs may overcome that hurdle because they bind to parts of the virus that remain constant even when it mutates. Germline targeting is an immune system-stimulating approach that guides naïve (precursor) B cells to develop into mature B cells that can produce bNAbs.
A class of bNAbs called 10E8 is a priority for HIV vaccine development because it neutralizes a particularly broad range of HIV variants. The 10E8 bNAb binds to a conserved region of the glycoprotein gp41 on HIV's surface involved in its entry into human immune cells.
Designing an immunogen—a molecule used in a vaccine that elicits a specific immune system response—to stimulate production of 10E8 bNAbs has been challenging because that key region of gp41 is hidden in a recessed crevice on HIV's surface. Prior vaccine immunogens have not generated bNAbs with the physical structure to reach and bind to gp41.
To address this challenge, the researchers engineered immunogens on nanoparticles that mimic the appearance of a specific part of gp41. They vaccinated rhesus macaque monkeys and mice with those immunogens and elicited specific responses from the 10E8 B cell precursors and induced antibodies that showed signs of maturing into bNAbs that could reach the hidden gp41 region. They observed similar responses when they used mRNA-encoded nanoparticles in mice.
The researchers also found that the same immunogens produced B cells that could mature to produce an additional type of gp41-directed bNAb called LN01. Finally, their laboratory analysis of human blood samples found that 10E8-class bNAb precursors occurred naturally in people without HIV, and that their immunogens bound to and isolated naïve human B cells with 10E8-like features.
Together, these observations suggest that the promising immunization data from mice and macaques has the potential for translation to humans.
According to the authors, these findings support the development of the immunogens as the first part of a multi-step vaccine regimen for humans. Their work further supports research in developing a germline-targeting strategy for priming the immune system to elicit a bNAb called VRC01. This bNAb was discovered by NIAID researchers almost 15 years ago. The goal of this line of research is to develop an HIV vaccine that generates multiple classes of bNAbs to prevent HIV.
More information: Torben Schiffner et al, Vaccination induces broadly neutralizing antibody precursors to HIV gp41, Nature Immunology (2024). DOI: 10.1038/s41590-024-01833-w