Genome surgery with CRISPR-Cas9 to prevent blindness
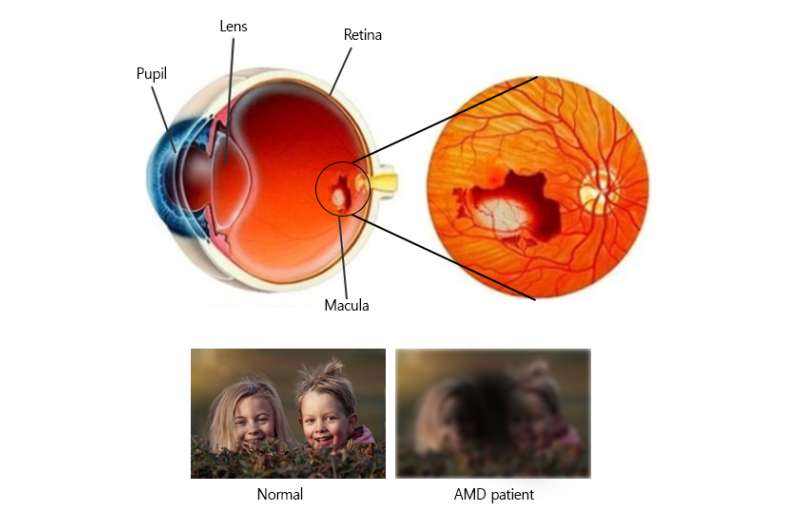
It is estimated that almost one in every ten people over 65 has some signs of age-related macular degeneration (AMD), and its prevalence is likely to increase as a consequence of the aging population. AMD is a form of blindness, common in Caucasians, which causes distorted vision and blind spots. Scientists at the Center for Genome Engineering, within the Institute for Basic Science (IBS) report the use of CRISPR-Cas9 in performing "gene surgery" in the layer of tissue that supports the retina of living mice. Published in Genome Research, this study combines basic research and mouse model applications.
The most common retinopathies causing blindness are 'retinopathy of prematurity' in children, 'diabetic retinopathy' and 'AMD' in older adults. In these diseases, abnormally high levels of the Vascular Endothelial Growth Factor (VEGF) are secreted. In AMD, VEGF causes the formation of new blood vessels in the eyes but also leads to leakages of blood and fluid into the eye, damaging an area at the center of the retina called macula.
Injections of anti-VEGF drugs are the most common treatment against AMD, but at least seven injections per year are required, because VEGF is continuously overexpressed by the cells of the diseased retinal pigment epithelium. Instead of such invasive treatments, IBS scientists believe that gene therapy with the third generation gene editing tool CRISPR-Cas9 could improve the situation. "The injections tackle the effects, but not the main cause of the problem. By editing the VEGF gene, we can achieve a longer-term cure," explains KIM Jin-Soo, Director of the Center for Genome Engineering.
CRISPR-Cas9 can precisely cut and correct DNA at the desired site in the genome. The CRISPR-Cas9 system works by cutting DNA at a target site, in this case, inside the VEGF gene. Two year ago, IBS scientists proved that a pre-assembled version of CRISPR-Cas9, a.k.a, Cas9 ribonucleoprotein (RNP), can be delivered to cells and stem cells to modify target genes. The pre-assembled complex works rapidly and degrades before the body has time to build up an immune response against it. Despite these advantages and previous successes, the difficulty in delivering pre-assembled CRISPR-Cas9 has limited its use in therapeutic applications.
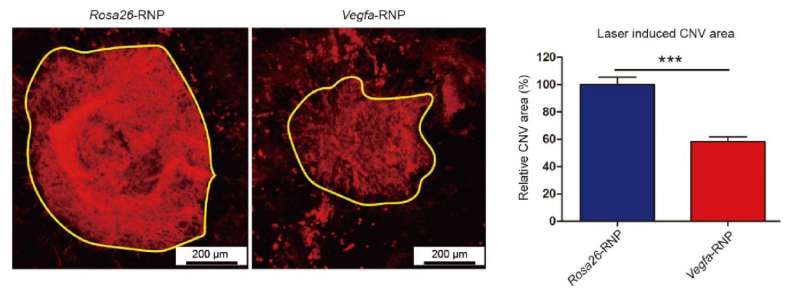
In this study, the research team successfully injected CRISPR-Cas9 into the eyes of a mice model with wet AMD and locally modified the VEGF gene. Initially they found that the delivery of the pre-assembled CRISPR-Cas9 complex is more efficient that the delivery of the same components in a plasmid form. Secondly, the complex disappeared after just 72 hours. Scientists assessed the whole genome of the animals and found the CRISPR-Cas9 complex modified only the VEGF gene and did not affect other genes. The progression of the eye disease was monitored by looking at choroidal neovascularization (CNV), the creation of new blood vessels between the retina and the sclera - a common problem of 'wet' macular degeneration - and researchers found the CNV area reduced by 58%. Moreover, a likely side effect, namely cone dysfunction, that takes only 3 days to show in these mice, did not occur a week after the treatment.
"We have developed a treatment to suppress CNV by inactivating the VEGF gene, one of the causes of AMD. We envision that, in the future, surgeons will be able to cut and paste disease-causing genetic elements in patients," explains Kim Jin-Soo.
While CRISPR-Cas9 is conventionally used to correct mutations causing hereditary diseases or cancer, this study suggests a new therapy for non-hereditary degenerative disease."We believe that this is a new therapeutic modality for the treatment of non-hereditary degenerative diseases," points out Professor KIM Jeong Hun (Seoul National University), "We confirmed the effect on the animal models of the disease and now we wish to continue with preclinical trials."
More information: Genome Research, DOI: 10.1101/gr.219089.116



















