FDA approves first-in-human trial for neural-enabled prosthetic hand system developed at FIU

Upper extremity amputees are one step closer to successfully picking up a cookie or an egg, thanks to a new advanced prosthetic system that is designed to restore sensation.
The U.S. Food and Drug Administration (FDA) has granted an investigational device exemption for the first-in-human trial with this technology. The system was developed at Florida International University by Ranu Jung and her Adaptive Neural Systems Laboratory team.
"The system is intended to restore the sense of touch, and hand opening which would allow users to precisely differentiate the size and fragility of various objects," said Ranu Jung, interim dean of the College of Engineering and Computing and a Wallace H. Coulter Eminent Scholars Chair in biomedical engineering. "The prostheses that exist today make it difficult for amputees to manipulate delicate and small objects because they can't feel them."
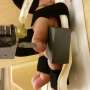
Jung's technology, a neural-enabled prosthetic hand system (NEPH), stimulates nerves in the arm to provide sensation as the person is using the prosthetic hand. The prosthetic system has the first fully implantable, wirelessly controlled Class-III device with electrodes that can be surgically implanted within the nerves of the residual arm.
"The system reflects an almost decade-long collaborative team effort. For the first time, amputees will be able to use a fully implantable, sensory enabled prosthetic hand system at home for daily activities and researchers will be able to assess the long term clinical impact of its use in real-world environments," said Jung.
Similar to a pacemaker, the system works by delivering small electrical pulses to specific nerves in the arm. Wires as thin as a hair strand are placed within nerve bundles in the arm and connected to an electrical stimulator. Sensors embedded in the prosthetic hand send signals wirelessly to the implanted stimulator, which then elicits sensation by delivering weak electrical pulses via the implanted electrodes. As a result, the person should be able to sense their hand opening position and grip items when the prosthetic hand encounters an object.
Participants in the trial will be able to use the system outside of the laboratory on a daily basis. At this time, the NEPH system is not approved by the FDA for U.S. commercial distribution, but if this first-in-human trial is successful, Jung and her team plan to continue testing through larger clinical trials and eventually seek such approval.

The system can potentially be interfaced with several different advanced prosthetic hands that are currently being developed commercially or under government support. Jung has spent the last decade developing the prosthetic hand system with the assistance of a Bioengineering Research Partnership grant from the National Institutes of Health (NIH), specifically its National Institute of Biomedical Imaging and Bioengineering (NIBIB) and Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD).
"This research journey started from foundational neuroscience and engineering principles to incorporating with painstaking accuracy the validation needed to make this system ready for clinical studies and this first-in-human trial. This unique system, integrating the long-term efforts of academia and industry, is an example of the bioengineering partnerships we promote," said Grace Peng, Ph.D., Program Director at NIBIB.
The HAPTIX program of the Defense Advanced Research Projects Agency (DARPA), which is an agency of the Department of Defense, is now supporting the first-in-human trial of the system. Florida International University, with Jung as principal investigator, will receive up to $2.2 million in support of this first-in-human trial.