New findings reverse hypothesis of GABA neurodevelopment in schizophrenia
New research by scientists at the University of Pittsburgh provides an unprecedented level of resolution and insight into disturbances in cortical GABAergic microcircuits, which are thought to underlie cognitive impairments in schizophrenia. Published in Biological Psychiatry, the study led by Dr. Kenneth Fish reveals new detailed understanding about alterations in neurocircuitry that point to abnormal neurodevelopment in the disorder.
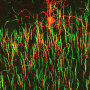
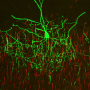
A recent generation of studies of postmortem brain tissue from people with schizophrenia, particularly from the laboratory of Professor David Lewis and his colleagues at the University of Pittsburgh, have shed light on schizophrenia-related abnormalities in the interplay of the main excitatory neurons, pyramidal neurons, and a specific class of inhibitory nerve cells, called chandelier cells, in the prefrontal cortex. Chandelier cells tune the activity of pyramidal neurons by releasing the inhibitory transmitter GABA through complex arrays of connections called cartridges. Prior postmortem studies have led to the hypothesis that most cartridges across cortical layers 2-5 have a decreased level of GABA reuptake, presumably a compensatory mechanism for lower GABA signaling associated with the disorder. However, the new postmortem study from the same group found evidence suggesting that the ability of chandelier cells to synthesize and release GABA within the prefrontal cortex is unaltered in schizophrenia. The study also demonstrates that the density of a specific subclass of chandelier cell cartridges is higher exclusively in layer 2 in the disease.
In the study, first author Brad Rocco, a graduate student in Fish's laboratory, and colleagues compared GABA synthesizing and packaging proteins within chandelier cell cartridges, as well as the density of these cartridges in the prefrontal cortex of 20 schizophrenia subjects and 20 comparison subjects. In layer 2, the density of cartridges arising from a transcriptionally-unique subset of chandelier cells containing calbindin was nearly 3-fold higher in the schizophrenia group. This subset only comprises a small fraction of neurons in the prefrontal cortex. In contrast, there was no difference in the density of chandelier cell cartridges lacking calbindin in layer 2 or in either type of cartridge across layers 3-6.
"These findings challenge prior studies that suggested that GABA deficits were a relatively universal feature of schizophrenia," said Dr. John Krystal, Editor of Biological Psychiatry.
The study was unable to determine the cause of this increased cartridge density, but the researchers suspect a developmental origin based on the layer specificity of the findings. Chandelier cell cartridges undergo dramatic pruning during development, and layer 2 matures much later than the deeper cortical layers, which had normal cartridge densities. "This finding suggests that the normal developmental pruning of these cartridges might be blunted in schizophrenia," said Fish.
According to Fish, substantial resources are being invested in the identification of transcriptionally-unique subtypes of human brain cells, an essential element in understanding the human brain, and the new findings highlight the importance of studying these types of cells in psychiatric disease. "The long-term goal of these efforts is to identify new ways to treat, cure, and even prevent brain disorders," said Fish.
More information: Brad R. Rocco et al, Alterations in a Unique Class of Cortical Chandelier Cell Axon Cartridges in Schizophrenia, Biological Psychiatry (2017). DOI: 10.1016/j.biopsych.2016.09.018