Treatment extends life for some with advanced prostate cancer
Adding a new anti-hormonal drug to the standard treatment for advanced prostate cancer has been shown to reduce the risk of dying by almost 40 percent, according to two studies published Saturday.
The drug, abiraterone, is sold by Janssen Pharmaceuticals under the brand name Zytiga.
According to one study, when combined with prednisone, which is typically given to men with metastatic prostate cancer, abiraterone reduced the risk of dying by 38 percent.
It also doubled the amount of time before the cancer worsened—from 14.8 to 33 months—said the findings released at the American Society for Clinical Oncology (ASCO) annual meeting.
The clinical trial included 1,200 patients in 34 countries and ran from February 2013 to December 2014.
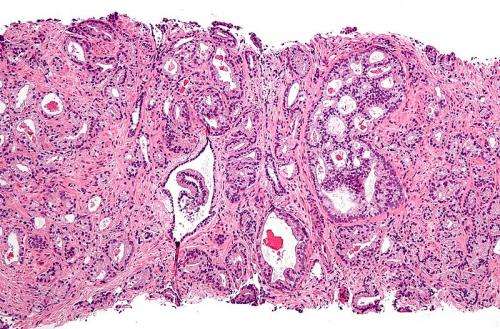
Most prostate cancers—92 percent of cases, according to the National Library of Medicine—are discovered early, before the tumors have spread elsewhere in the body.
But for the five percent or so of men who are diagnosed with prostate cancer that has already spread, the outlook can be dire.
"There is a large unmet need to improve treatment for men with newly diagnosed metastatic cancer, who die of the disease within less than five years on average," said lead study author Karim Fizazi head of the Department of Cancer Medicine at Gustave Roussy, University Paris-Sud in Villejuif, France.
Prostate cancer is the leading cause of death among men in the United States, with more than 161,000 new cases diagnosed each year and more than 26,000 deaths, according to the American Cancer Society.
Hormone-fueled tumors
A second clinical trial involving nearly 2,000 men showed that adding abiraterone to a standard initial treatment regimen for high-risk, advanced prostate cancer lowered the relative risk of death by 37 percent.
The study was the largest to date on abiraterone as first-line therapy for advanced prostate cancer, in combination with standard therapy.
"Abiraterone not only prolonged life, but also lowered the chance of relapse by 70 percent and reduced the chance of serious bone complications by 50 percent," said lead study author Nicholas James, professor of clinical oncology at Queen Elizabeth Hospital in Birmingham.
"Based on the magnitude of clinical benefit, we believe that the upfront care for patients newly diagnosed with advanced prostate cancer should change."
The hormone testosterone fuels the growth of prostate cancer cells.
Men with prostate cancer are commonly treated with androgen deprivation therapy, which slows the cancer growth by preventing testicles from making testosterone and other similar hormones—or androgens.
Even when men are on ADT, other organs in their bodies may continue to make small amounts of testosterone and other androgens.
Experts say abiraterone boosts standard therapy because it stops production of both testosterone and other androgens throughout the body.
Side effects
However, experts warned that severe side effects were more common in patients who took abiraterone.
The main side effects were cardiovascular problems such as high blood pressure, and abnormal liver enzymes.
In James' study, severe side effects occurred in 41 percent of patients compared to 29 percent of patients in the standard therapy group.
In Fizazi's, 20 percent of patients in the combination therapy group reported high blood pressure, compared to 10 percent on standard therapy alone.
"We need to be cautious when using abiraterone in men who have an increased risk for heart problems, such as those with diabetes," said Fizazi.
Overall, the treatment appears to mark a step forward against advanced prostate cancer, said Sumanta Kumar Pal, an ASCO expert who was not involved in the study.
"This is good news because using abiraterone could help many people live longer with fairly few additional side effects."
© 2017 AFP